Cardiovascular devices
on Chest X-Ray
Frederieke Elsinger, Robin Smithuis and Anje Spijkerboer
Vancouver General Hospital in Canada, Alrijne hospital in Leidenand the Amsterdam University Medical Center, the Netherlands
Publicationdate
The number of implanted cardiovascular devices has dramatically increased in recent years.
With the advent of implantable cardioverterdefibrillators and cardiac resynchronization therapy, devices have become more complex.
There is also an increase of minimally invasive procedures to replace heart valves.
An overview is presented of common cardiovascular devices and procedures.
Radiology plays an important role in the initial assessment and follow-up of these devices.
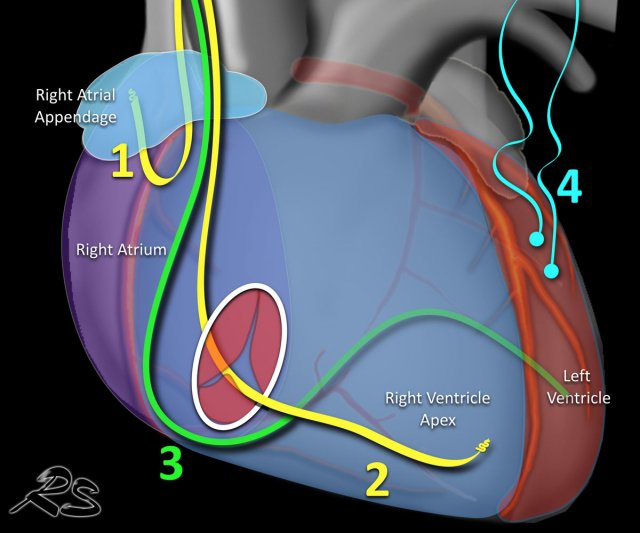
Pacemakers
There are two types of cardiac conduction devices (CCD's):
- Pacemakers that pace the heart during periods of bradycardia.
- Inplantable cardioverter-defibrillators (ICD's) that are capable of defibrillating the heart to prevent cardiac arrest.
Pacemakers can have leads, that pace:
- Right atrium - tip of lead is located in atrial appendage.
- Right ventrical - tip in apex.
- Left ventricle - lead through the coronary sinus ends in posterior cardiac vein, used for cardiac synchronization therapy in patients with bundle branch blocks.
- Epicardial - placed on ventricle mainly used in cardiac surgery.
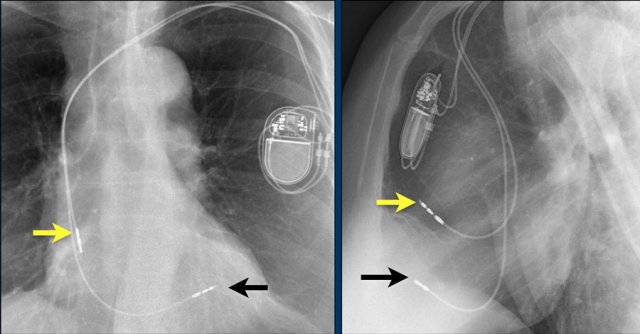
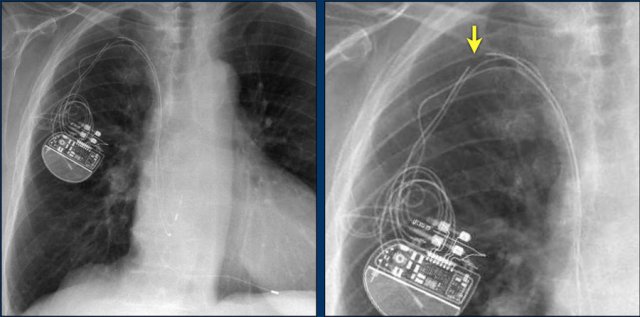
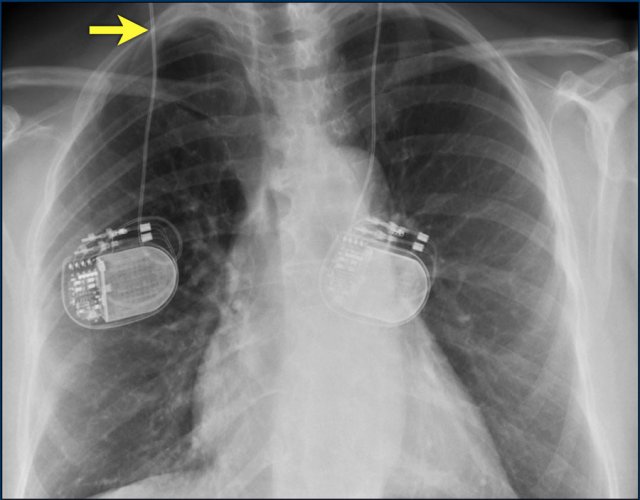
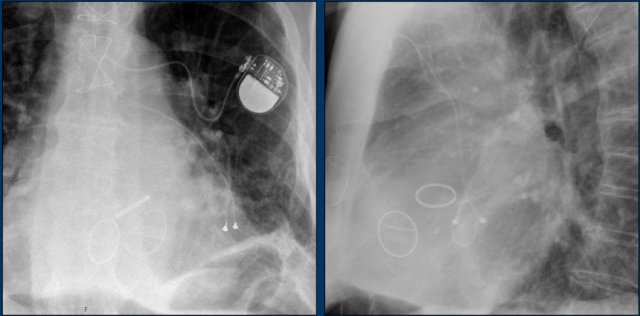
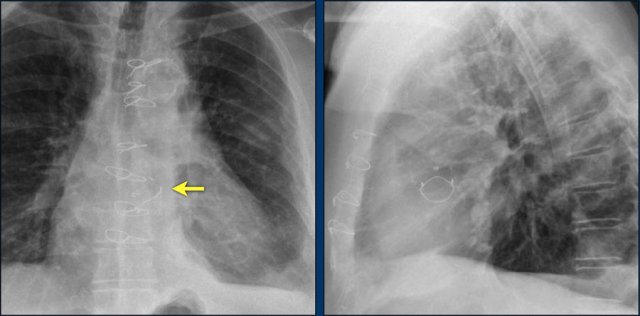
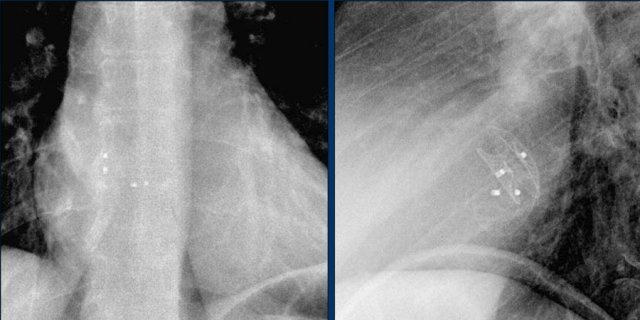
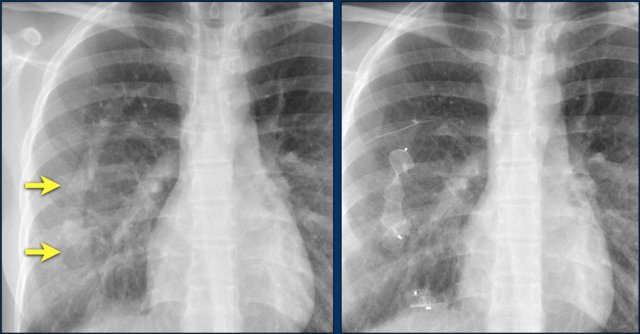
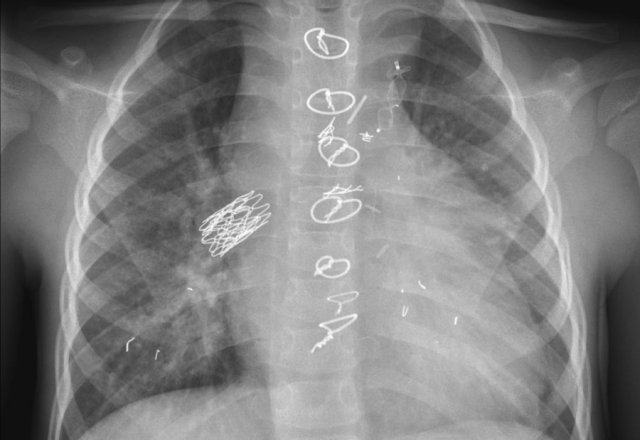
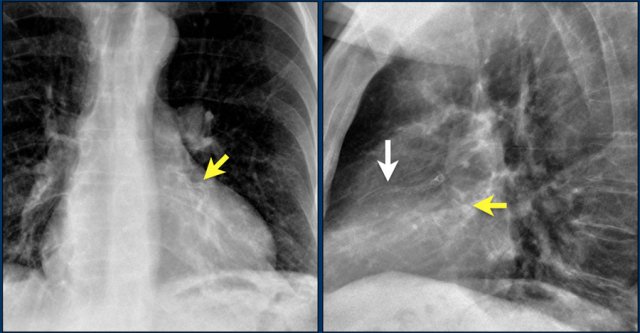
The images show a pacemaker with an atrial and ventricular lead.
The tip of the atrial lead is pointed upward and anteriorly, because the ideal position is in the right atrial appendage, where it is anchored within the coarse trabeculae.
The tip of the ventricular lead is positioned in the apex of the right ventricle, which is located to the left of the spine on a frontal chest X-ray and anteriorly on a lateral view.
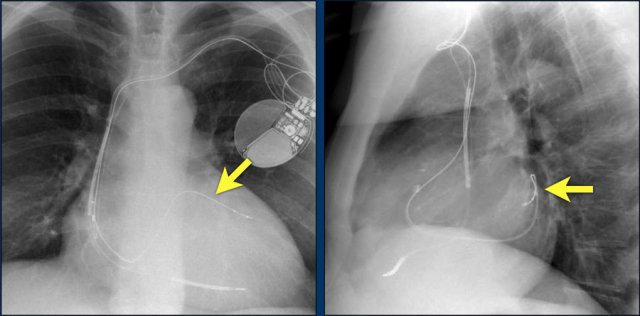
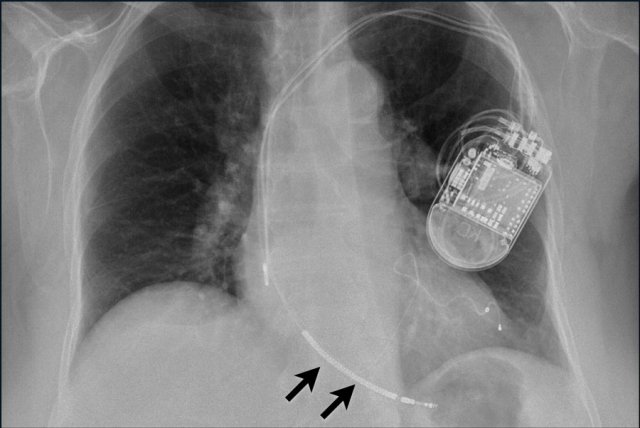
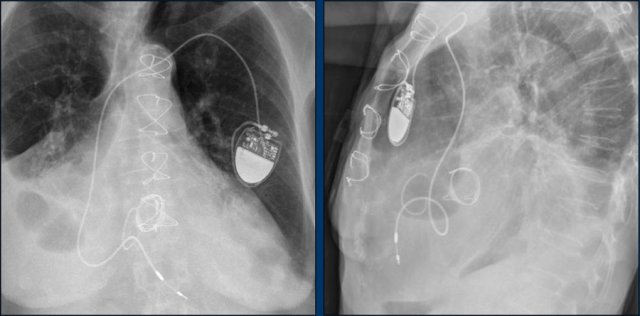
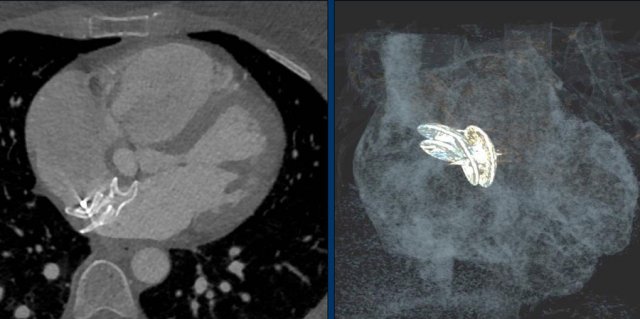
Here an example of an ICD with wires in the right atrial appendage, the apex of the right ventricle and a lead to the left ventricle in the posterior coronary vein.
You can recognize the two shock coils of the ICD as thicker white bands along the course of the lead.
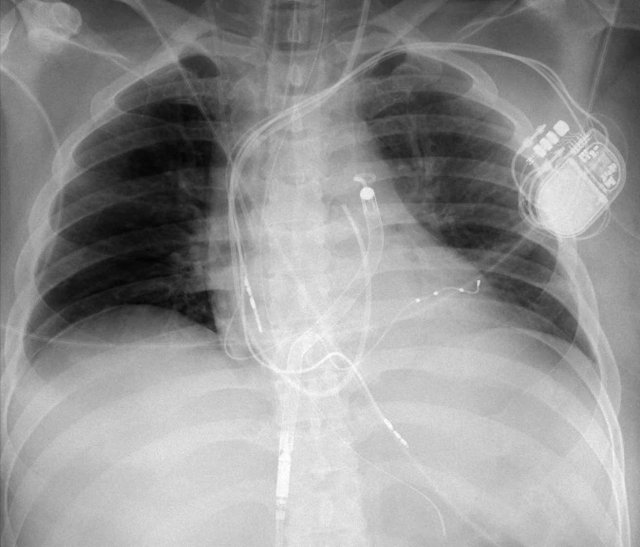
Cardiac Resynchronization Therapy
Cardiac resynchronization therapy with a biventricular pacer is performed to synchronize the contraction of the right and left ventricle in patients with severe systolic heart failure and left or right bundle branch blocks or other intraventricular conduction defects.
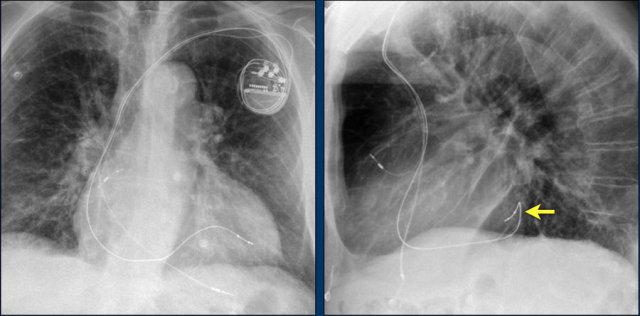
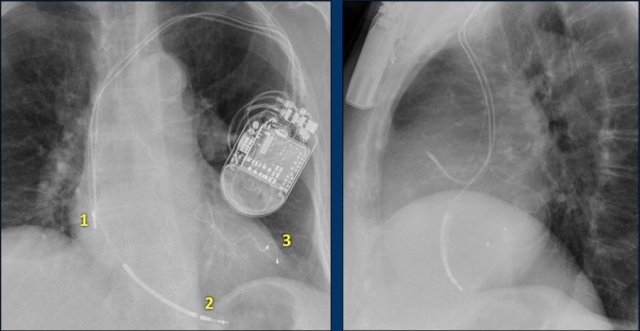
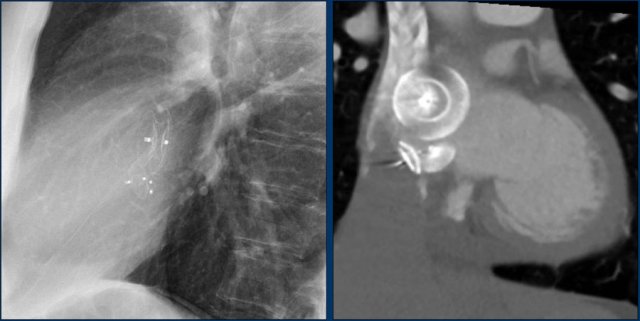
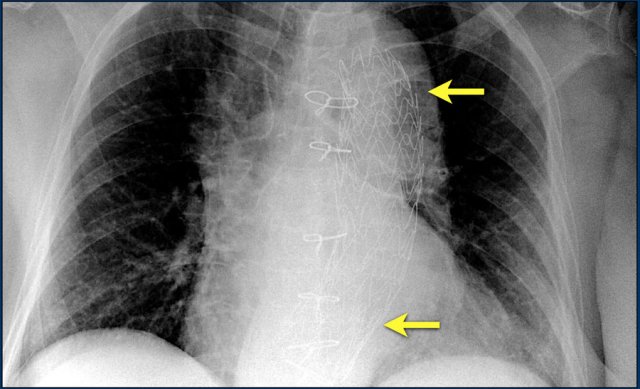
Here a biventricular pacemaker with three leads.
The right atrial pacemaker lead is located in the atrial appendage.
This lead first travels inferiorly into the right atrium and then turns upward and anteriorly where it is anchored within the trabeculae of the atrial appendage.
The left ventricular lead travels through the right atrium and the sinus coronarius and is finally positioned posteriorly into a cardiac vein on the left.
Epicardial pacing leads
Epicardial leads are frequently placed during cardiac surgery in order to allow cardiac pacing post-surgery.
Some centers place them in all cardiac surgery patients, while others only in those who have rhythm disturbances intra-operatively.
After a few days they can be removed by simple traction.
Sometimes they are left in place.
Retained epicardial wires do not seem to present a hazard to patients in the MR environment.
However, this conclusion applied mostly to non-cardiac MR examinations.
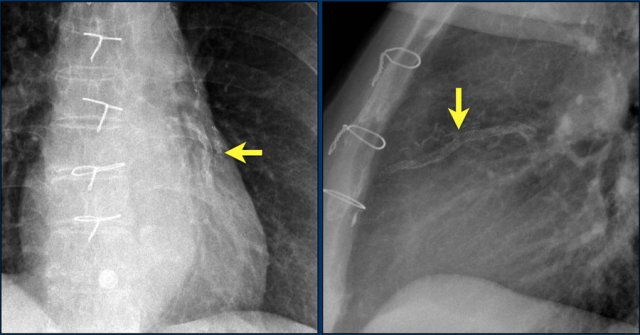
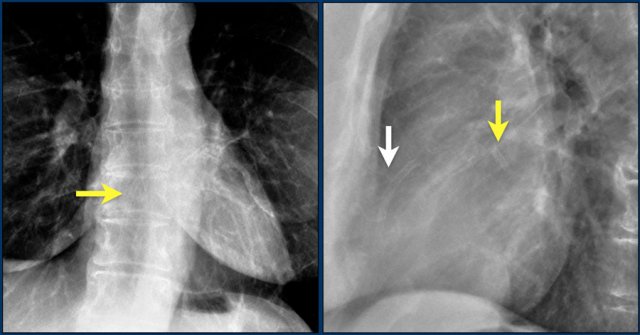
First study the images.
Then continue reading.
The findings are:
- Two epicardial leads connected to pacemaker
- ICD
- two leads to right ventricular apex
- one lead contains two shock coils
- tricuspid valve (arrows)
- mitral valve
Pacemakers can have various complications:
- Pneumothorax
- Lead fracture
- Lead dislocation
- Infection
- Perforation
- Twiddler's syndrome
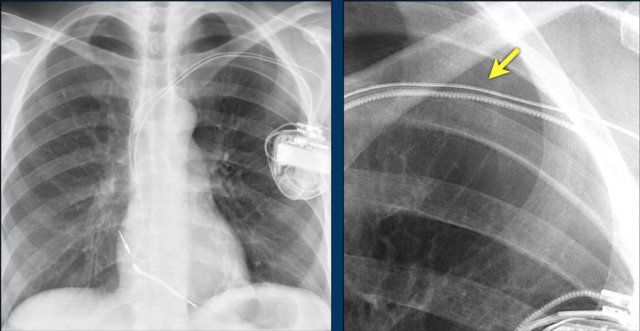
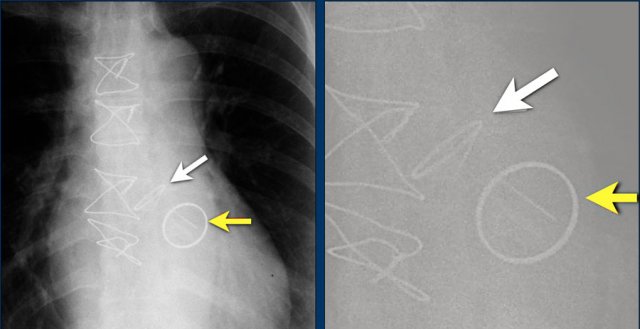
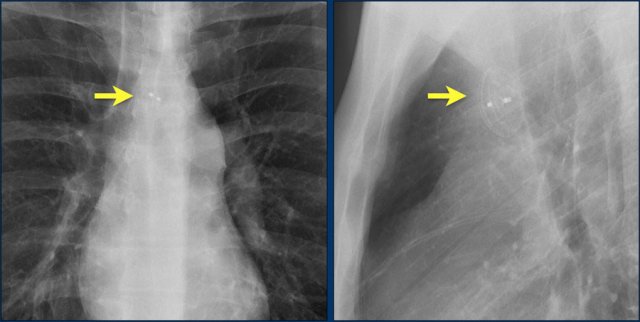
The image is taken immediately after placement of an ICD.
There is a small pneumothorax (arrow).
This is the most common complication.
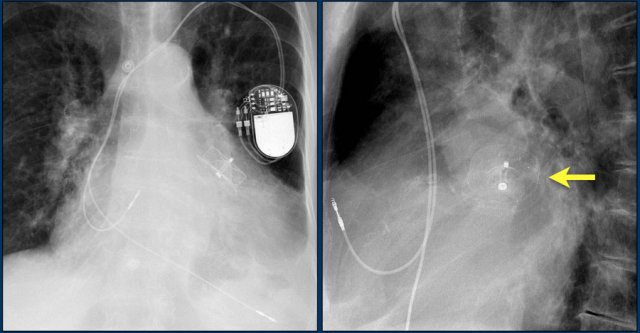
Aberrant lead position
This patient has a persistent left-sided superior vena cava (LVCS).
This is a rare congenital anomaly that may be found at the time of cardiac device lead insertion.
The persistent LVCS communicates with the right atrium through the coronary sinus.
The transvenous placement of cardiac device lead via persistent LSVC can be technically difficult and in some cases not possible.
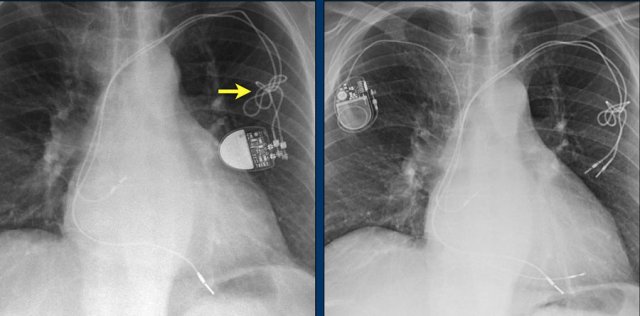
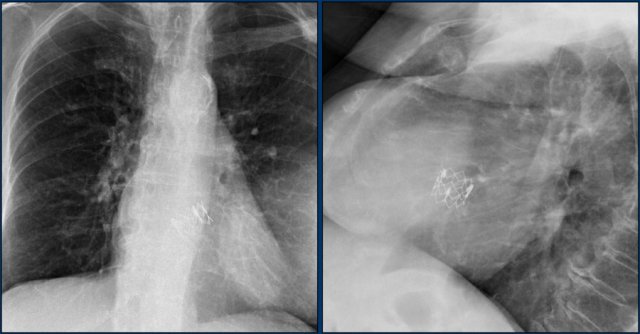
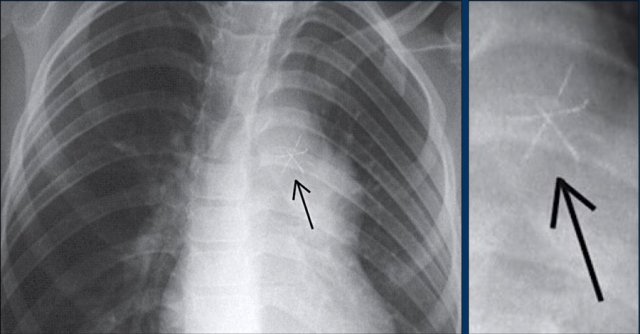
Lead fracture
Obvious fracture of one of the leads.
Subtle lead fracture in malfunctioning pacemaker.
Extremely subtle fracture line (arrow).
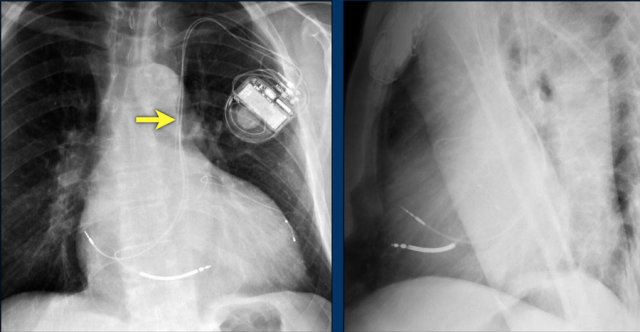
Retained leads
Here a patient with an ICD with one lead and two shock coils.
There is an retained lead still visible of an old pacemaker that was removed.
Many patients have endocardial pacemaker leads left in place after pulse generator removal.
The safety of MR in patients with retained endocardial pacemaker wires has not been systematically investigated to date.
However, due to the potential threat that they may act as "antennas" with significant heating - it is not recommended to scan those patients (reference).
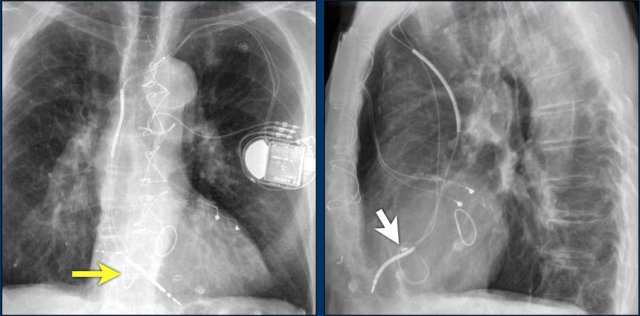
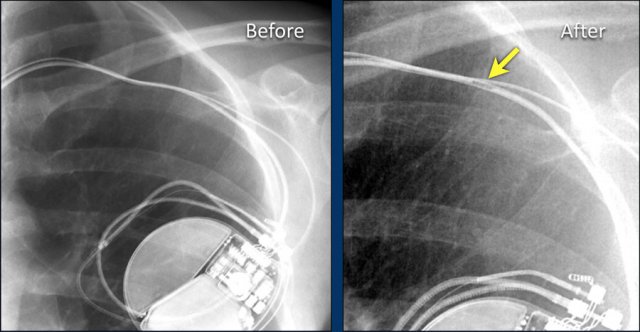
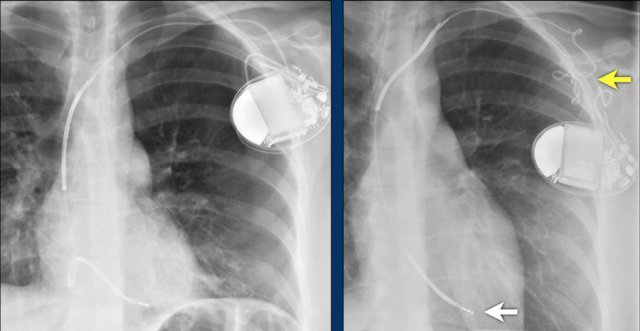
Twiddler's Syndrome
Twiddler's syndrome is a malfunction of a pacemaker due to the patient's manipulation of the device and the subsequent dislodging of the leads.
This results in malfunctioning of the pacemaker or sometimes stimulation of other structures like phrenic nerve or brachial plexus.
Here a patient with a normally placed ICD on the left image.
Months later there was malfunction due to ICD box and lead rotation (yellow arrow) and retraction (white arrow).
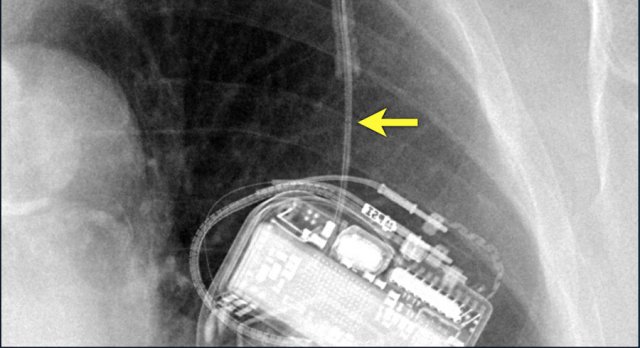
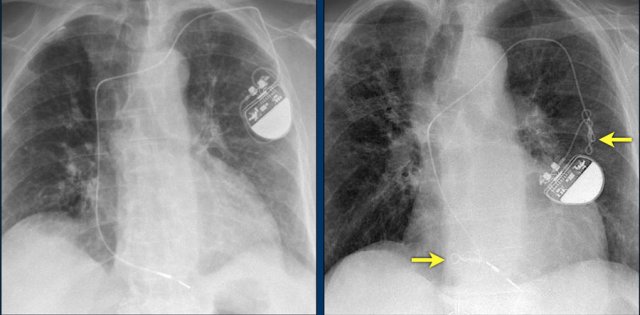
Here another patient with the Twiddler's syndrome.
Notice curling of the lead near the pacemaker and at the tip in the right ventricle (arrows).
Here another patient with Twiddler.
There was malfunction of the pacemaker.
A new pacemaker was inserted on the right side.
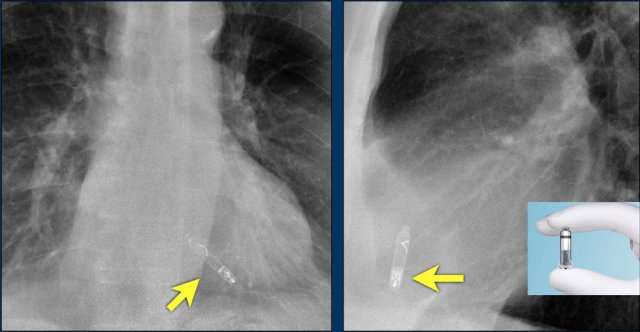
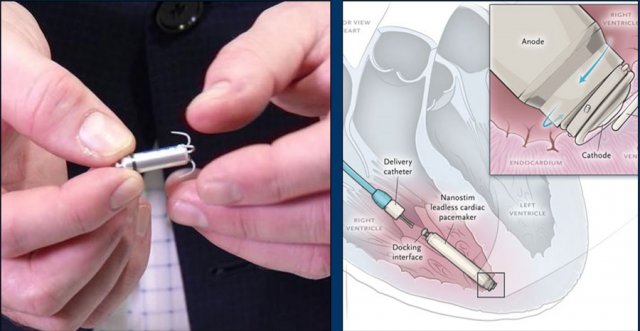
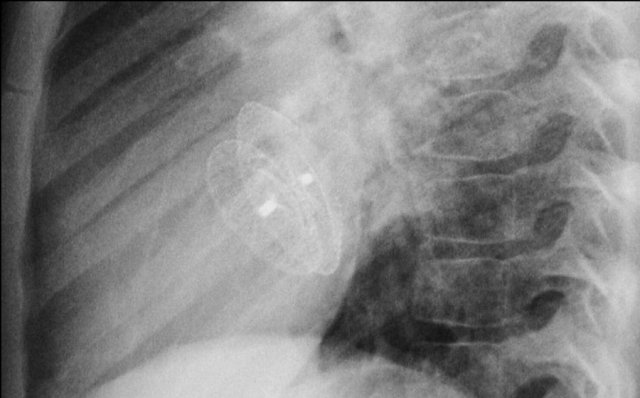
Wiireless pacemaker device
A Micra device is a small wireless pacemaker device, that is transfemorally implanted in the apex of the right ventricle.
Close up view of the Micra device.
Brain stimulators
Parkinson brain stimulators have similar generators as cardiac pacemakers and are also placed in the subcutaneous chest.
They should not be confused with pacemakers.
Implantable cardioverter-defibrillators
Implantable cardioverter-defibrillators or ICD's are devices that can recognize ventricular tachycardia and fibrillation and terminate it by delivering an electrical shock.
ICD's are implanted in patients with cardiomyopathy and a low left ventricular ejection fraction because they are at risk of ventricular tachycardia, ventricular fibrillation and sudden cardiac death.
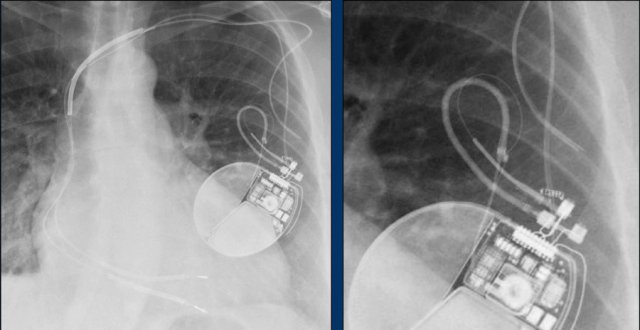
This patient has a single coil ICD system (figure).
The arrows point to the shock coil.
Continue with next images.
This patient has three leads:
- Right atrial lead
- Right ventricular lead (with shock coil)
- Left ventricular lead in coronary sinus and cardiac vein
Here another patient with an ICD with only one lead and one shock coil.
Here an ICD with one lead and two shock coils.
Subcutaneous ICD
The S-ICD or subcutaneous implantable cardioverter-defibrillator is not connected to the heart or the vessels.
A presternal wire is placed and connected to a laterally placed ICD with the heart located between the wire and the box.
This system is not capable of pacing the heart.
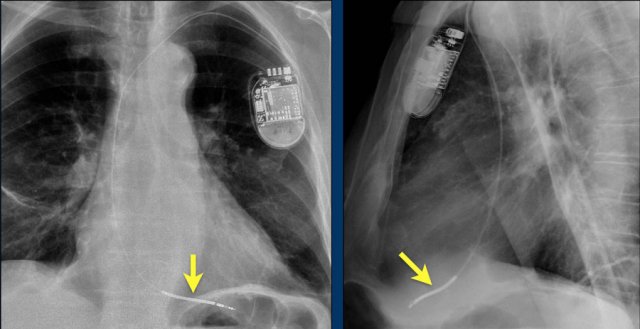
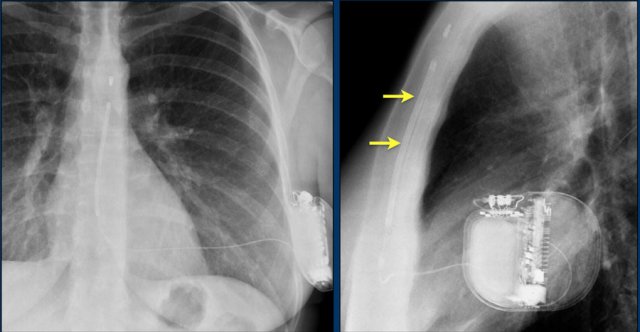
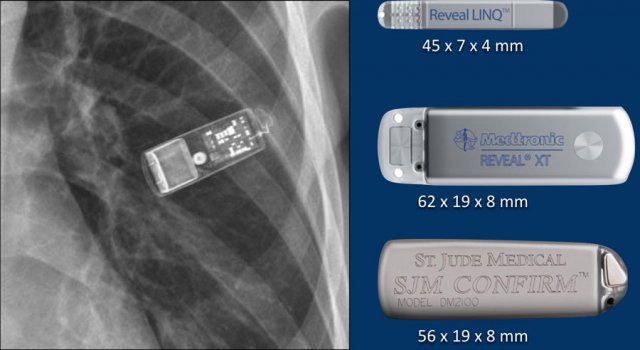
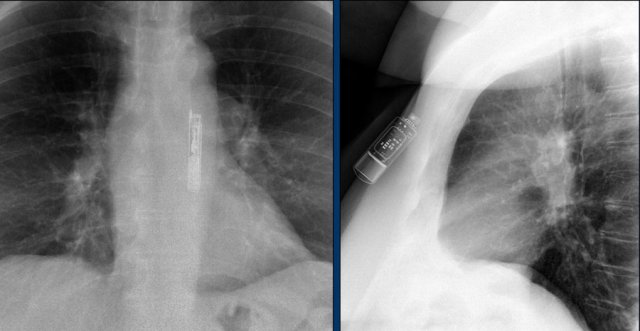
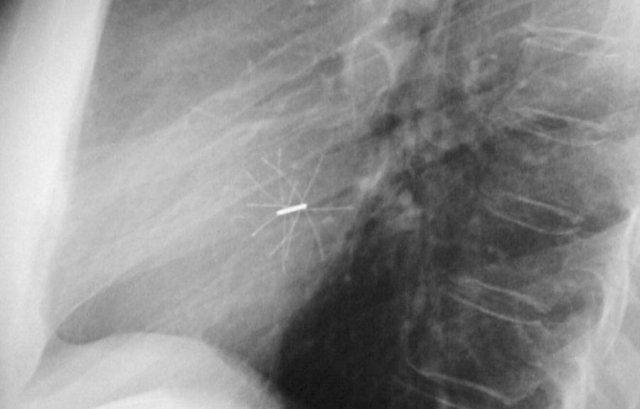
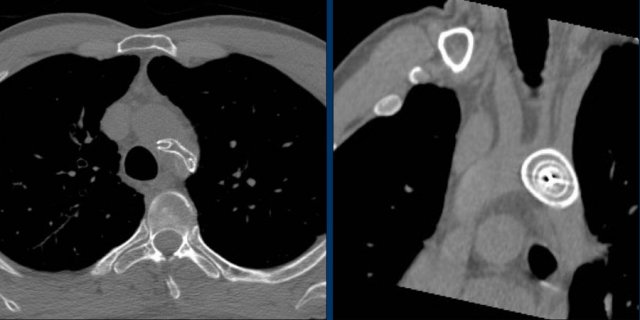
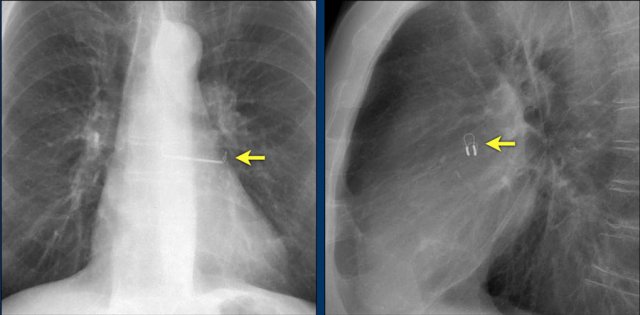
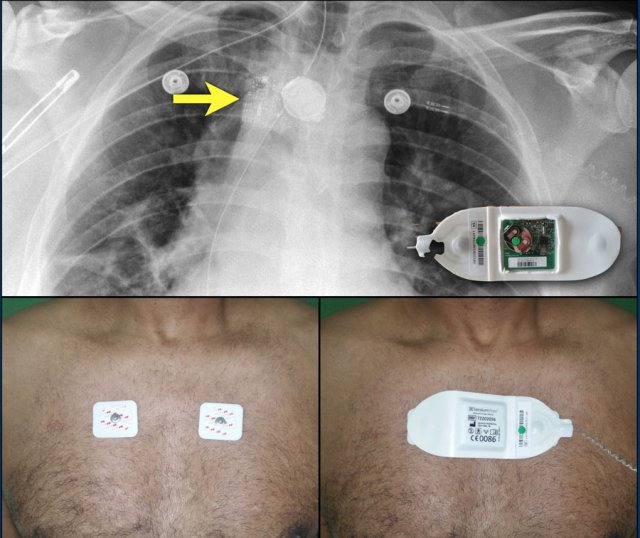
Loop recorders
Loop recorders are implantable cardiac monitors to continuously record the cardiac rhythm in patients with unexplained palpitations or syncope.
They are placed subcutaneously.
These devices are getting smaller and smaller and should not be mistaken for an USB flashdrive.
Loop recorders have a variety of uses including monitoring patients with unexplained syncopal episodes and assessment of patients with atrial fibrillation, ventricular arrhythmias, or conduction disturbances.
These recorders can provide more than a year of monitoring that may be activated by the patient or be automatically activated.
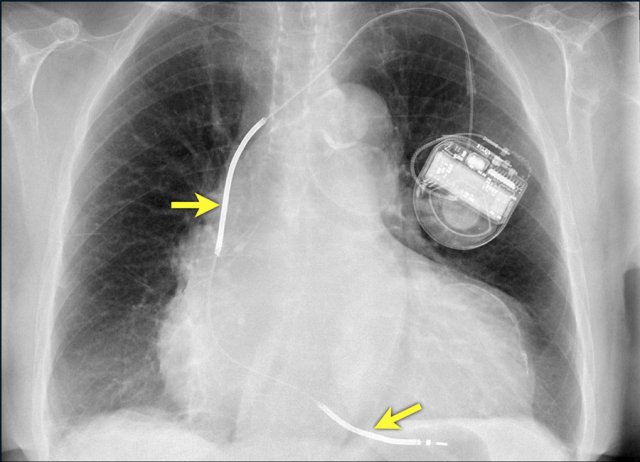
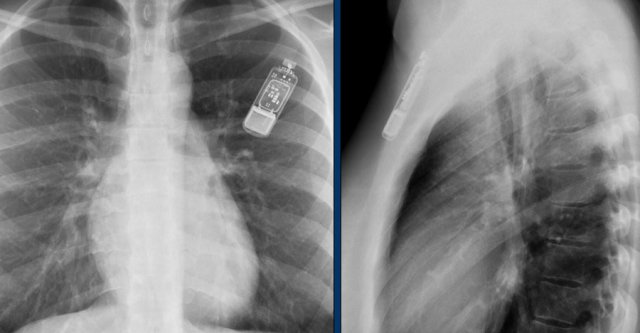
Another example of a loop recorder.
Valve replacement and repair
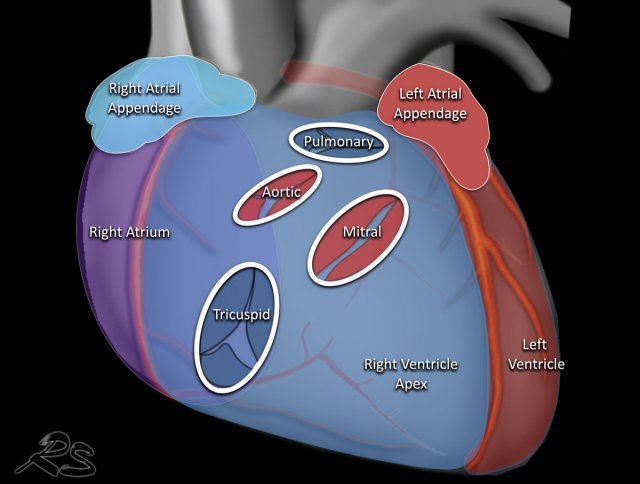
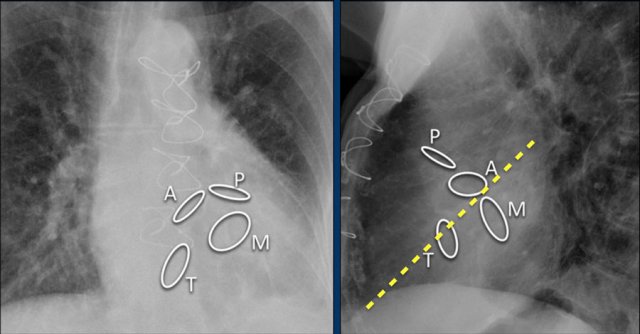
This is an illustration of the heart valves in the coronal plane.
The heart valves are best determined on a lateral radiograph.
The aortic and pulmonary valves are above a line drawn from the apex to the base of the heart, while the mitral and tricuspid valves are below that line
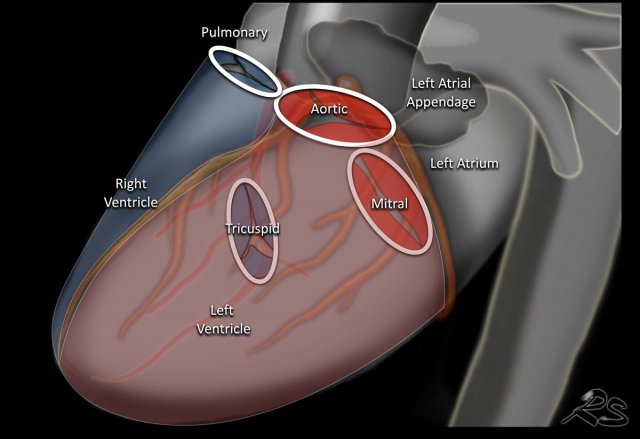
Here we see the normal valve position on an PA and lateral chest X-ray.
On the lateral chest film the aortic and pulmonary valves are located above the line from base of the heart to the apex and tricuspid and mitral valves are below this line.
Different types of valves:
- Mechanical or prosthetic valves
These are from synthetic material.
These patients are on anticoagulant therapy. - Biological or bioprosthetic valves
These are made from animal valves. No anticoagulation therapy needed
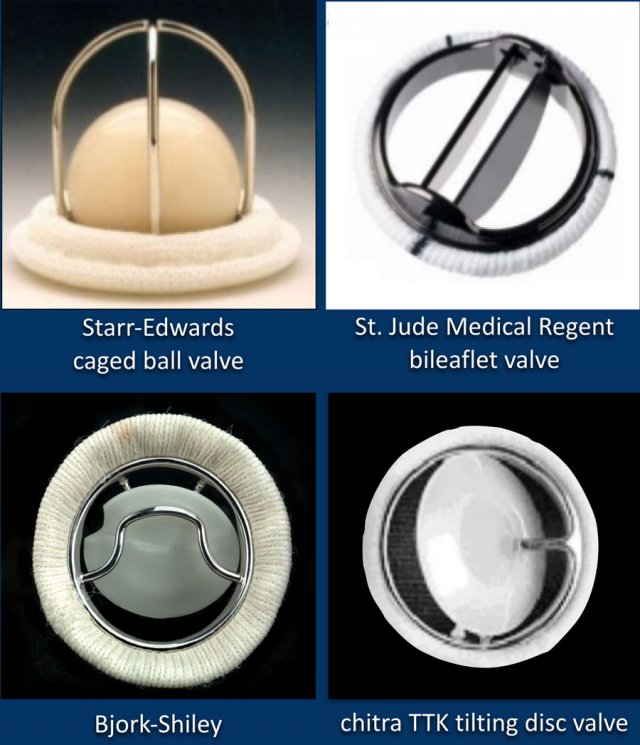
Mechanical valves
The images show some examples of mechanical heart valves.
These are manufactured mechanical valves.
They last for about 20 years.
Patients require anticoagulantia and therefore their use is decreasing.
The St. Jude bi-leaflet mechanical valve is most commonly used and has a radiopaque peripheral ring.
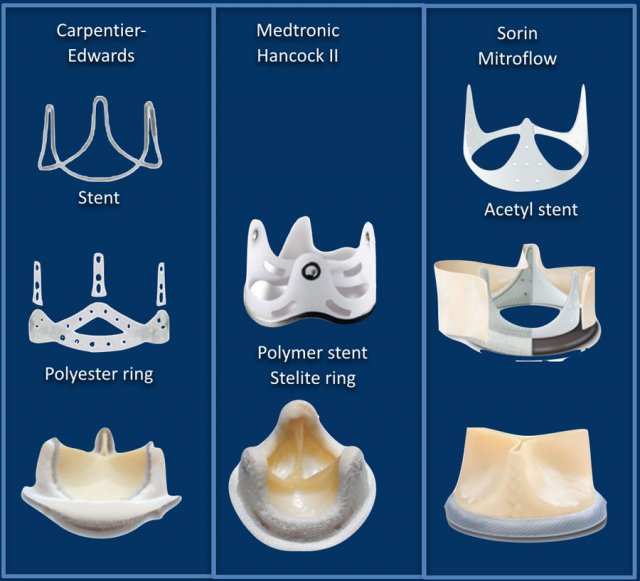
Bioprosthetic valves
Here some examples of prostetic heart valves.
The main limitation of bioprosthetic valves is their limited durability, which exposes the patient to the risk of valve reintervention.
Last about 10-15 years but do not require anticoagulation.
Typically chosen for older patients, those with a contraindication to anticoagulation or for lower velocity inflow valves (mitral and tricuspid).
Aortic valve prosthesis
in good position
Tricuspid valves
Tricuspid stenosis is the result of rheumatic heart disease and is treated with valve replacement.
Tricuspid regurgitation is the result of right ventricular dilatation and is treated with annuloplasty.
This patient has three valves repaired:
- Mitral
- Tricuspid
- Aortic
There is a pacemaker with epicardial leads.
This was done, because it was thought that a normally placed lead to the right ventricle would interfere too much with the function of the tricuspid valve prosthesis.
White arrow points to aortic valve.
Yellow arrow points to mitral valve.
TAVR
TAVR is a transcatheter aortic valve repair.
The stenotic aortic valve is repaired by placing a prosthetic valve within the diseased valve.
TAVR is for patients who have a high risk for an open procedure.
The procedural success rate is 90%.
TAVR is associated with higher rates of vascular injury, paravalvular regurgitation and the need for permanent pacemaker placement.
Pulmonary valves
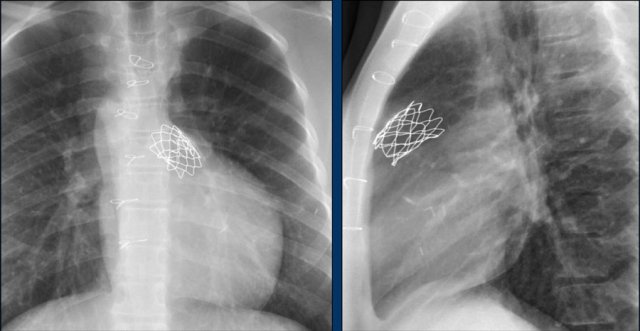
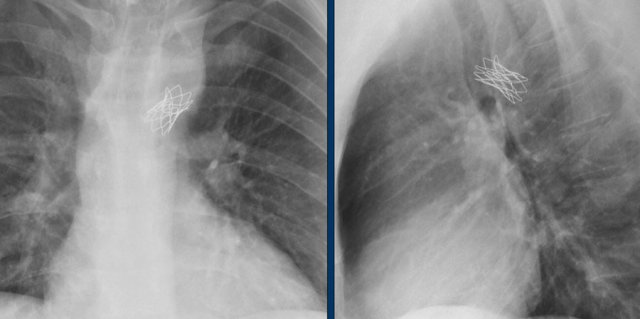
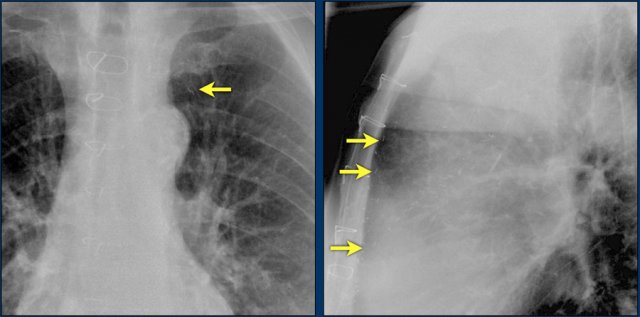
The Melody valve is a transcatheter pulmonary valve (TPV).
It is a bovine jugular vein sewn within a platinum-iridium stent.
The Melody valve is inserted through a catheter percutaneously.
This Melody stent has multiple fractures (yellow arrows)
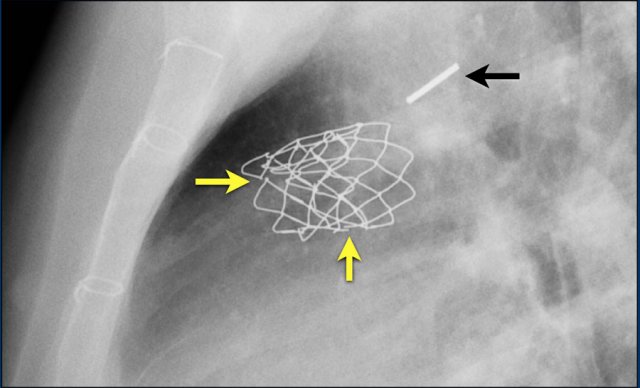
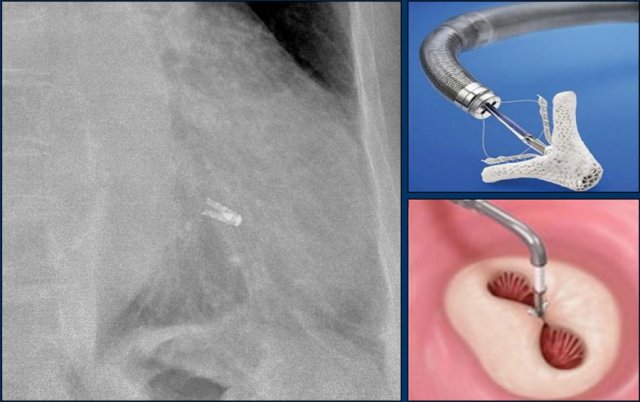
MitraClip
In patients with severe mitral regurgitation and who are not a candidate for open heart surgery, a transcatheter mitral valve repair, known as the MitraClip, is an appropriate therapy.
The centre of the mitral leaflets are approximated by the Mitraclip to reduce the regurgitation while still leaving enough room for passage of blood from the left atrium to the left ventricle (figure).
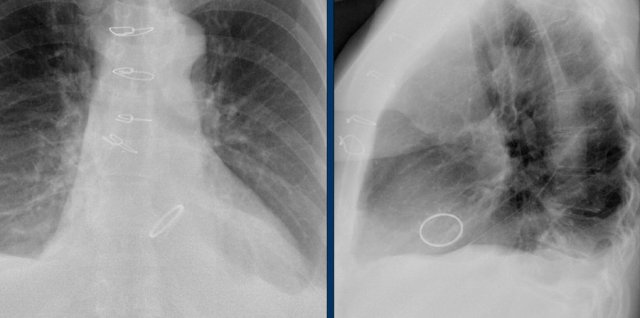
Here a patient with three different valves: aortic, mitral and tricuspid.
Aortic valve replacement.
This patient has a Medtronic Hancock mitral valve replacement and an annuloplasty of the tricuspid valve.
Closure devices
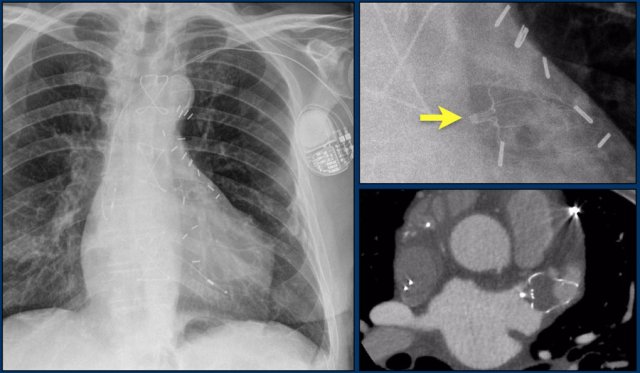
Atrial Septal defect
Closure devices are used in patients with ASD and signs of right ventricle overload which can lead to heart failure and atrial fibrillation.
Closure devices are often self-adjusting and lie flat against the atrial or ventricular septum.
This flat design helps create a natural profile.
Here a lateral view of a child with an ASD and an Amplatz closure device.
A lateral view of a child with an ASD and an Umbrella Rashkind closure device.
Images of a patient with an ASD and an Amplatz closure device.
Here a patient with a larger defect, that was closed with two devices.
Continue with the CT-images.
Transverse CT-image and coronal reconstruction of the same patient.
This patient has three closure devices.
Dislocation of the closure device.
CT demonstrates, that the Amplatz device is dislocated into the aortic arch.
First look at the images.
Then continue reading.
This is an Amplatz cardiac plug to close the left atrial appendage.
This is an alternative to oral anticoagulation for prevention of thromboembolic stroke in patients with non-valvular atrial fibrillation
Watchman
A commonly used left atrial appendage closure device is the Watchman implant.
It is a preventive measure for stroke caused by clots originating from the heart in patients with atrial fibrillation for which anticoagulants are contraindicated.
Patent Ductus Arteriosus
A patent ductus arteriosus (PDA) is a persistent communication between the descending thoracic aorta and the pulmonary artery that results from failure of normal physiologic closure of the fetal ductus.
During the first 60 hours of life, spontaneous closure of the ductus occurs in 55% of full-term newborn infants.
By 2-6 months of age, closure occurs in more than 95% of healthy infants.
Large PDA in older children and adults can lead to pulmonary hypertension and chronic heart failure as a result of a left-to-right shunt.
These are images of a patient with a persistent ductus arteriosus.
The ductus was closed with an Amplatz plug device.
Scimitar vein plugging
Scimitar syndrome is characterized by partial anomalous pulmonary venous return, in which an abnormal right pulmonary vein drains into the inferior vena cava.
Correction is indicated in adults with signs of pulmonary volume overload or right heart dilation.
Images:
Scimitar vein pre- end post plugging.
Vascular Stents
Coarctation
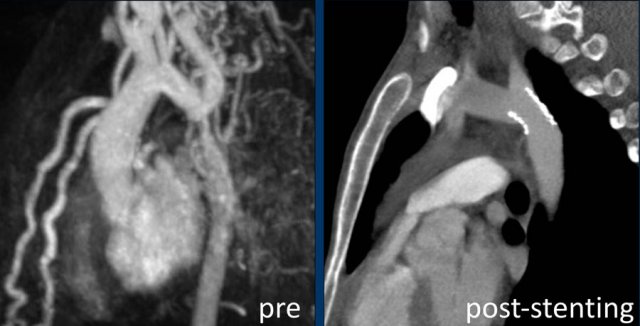
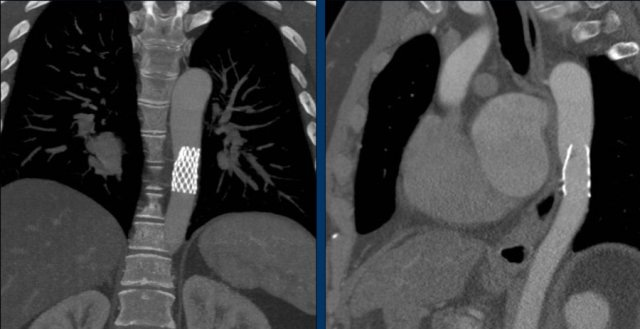
Intravascular stent therapy is considered a primary therapeutic option for most adults and adolescents with coarctation of the aorta (refence).
Image: intravascular stent in good position.
Continue...
MRA-image of the same patient pre-stenting.
CTA-image post-stenting.
Image: dislocated stent.
..
TEVAR
Thoracic EndoVascular Aortic Repair (TEVAR) is a procedure that involves placement of a covered stent in the aorta in patients with a symptomatic thoracic aneurysm.
The stent graft is delivered in a collapsed state through a catheter, that is most often inserted into the femoral artery and positioned using X-ray guidance.
The stent graft is then expanded to span and cover the site of aortic injury (reference).
Coronary stents
X-rays show a stent projecting over the anterior ventricle wall, where the LAD is located.
First look at the images.
Then continue reading.
Where are the stents positioned?
The coronary stents are positioned in the right coronary artery (yellow arrow) and the left anterior descending artery (white arrow).
First look at the images.
Then continue reading.
Where are the stents positioned?
The coronary stents are positioned in the left circumflex artery (yellow arrow) and the left anterior descending artery (white arrow).
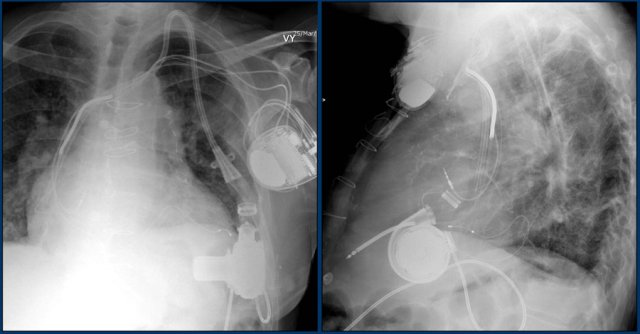
Left ventricular assist device
A left ventricular assist device (LVAD) is a surgically implanted device that takes over ventricular pump function in patients with severely impaired ejection fraction.
They were initially implanted as a bridge to cardiac transplant or during myocardial recovery, but are now also used in patients not suitable for transplant.
Here we see a patient with a LVAD.
The outflow cannula is inserted in the apex and the radiolucent outflow cannula is connected to the ascending aorta.
Also note the triple lead, double shock coil ICD and previous LIMA graft clips.
Impella
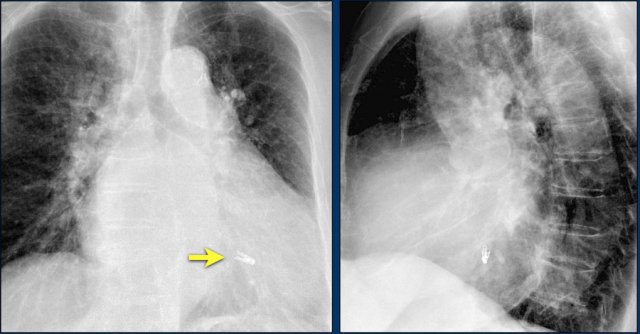
An Impella is a small, temporary heart pump, attached to a transfemoral (or axillary) catheter.
It is intended for short term use (from 6 hours to < 14 days, depending on the type) during high risk PCI or in patients recovering from AMI cardiogenic shock.
The Impella’s inlet is placed in the left ventricle, where blood is apirated and delivered to the outlet which is situated in the ascending aorta.
The Chest X-Ray shows correct placement of impella with inlet (yellow arrow) and outlet (white arrow) in the left ventricle and ascending aorta.
In this video you can watch how the impella works.
The Impella can also be used to offload the right ventricle.
The current installations include:
- Central venous catheter in the right internal jugular vein
- Feeding tube positioned in the stomach.
- Endotracheal tube placed above the carina.
- 3-lead pacemaker is present with electrodes in the right ventricle (RV), right atrium (RA), and left ventricle (LV).
- Swan-Ganz catheter located in the pulmonary artery.
- Right-sided Impella device located in the pulmonary artery used to offload the right ventricle in the setting of right-sided heart failure.
- EKG electrodes in place.
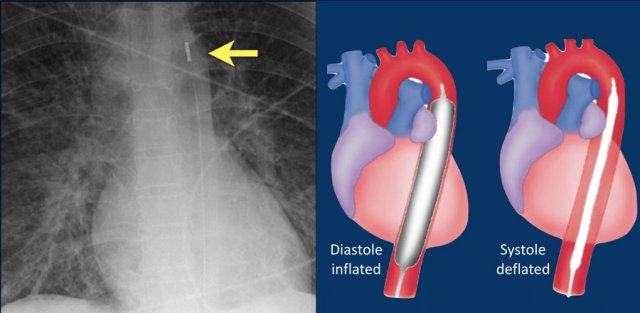
Intra-aortic balloon pump
The intra-aortic balloon pump (IABP) is a mechanical device that increases myocardial oxygen perfusion and indirectly increases cardiac output through afterload reduction.
It consists of a cylindrical polyurethane balloon that sits in the aorta, approximately 2 centimeters (0.79 in) from the left subclavian artery.
The balloon inflates and deflates via counter pulsation, meaning it actively deflates in systoleand inflates in diastole.
Systolic deflation decreases afterload through a vacuum effect and indirectly increases forward flow from the heart.
Diastolic inflation increases blood flow to the coronary arteries via retrograde flow.
These actions combine to decrease myocardial oxygen demand and increase myocardial oxygen supply (reference).
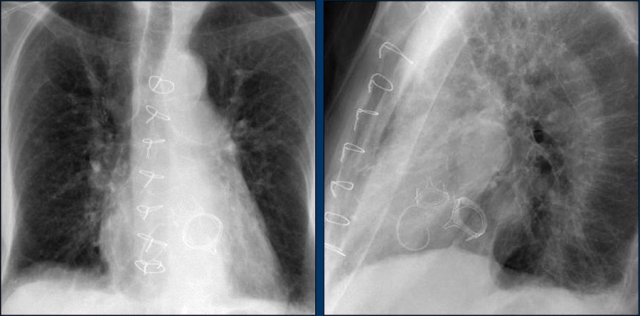
Vascular surgery clips - CABG
The standard bypass operation involves LIMA-to-LAD graft combined with vein grafts.
LIMA stands for Left internal mammary artery bypass graft.
The distal LIMA is anastomosed with the post-stenotic LAD and surgical clips extend from the apex of the aortic arch (image).
RIMA grafts are used less frequently because of lower patency and because the RIMA is shorter and usually does not reach the target arteries.
The left IMA is separated from the chest wall and connected to the coronary target vessel distal to the stenosis.
The radiograph shows the vascular clips of a left and right IMA CABG.
Mini Maze procedure
The Mini Maze procedure is a minimally-invasive ablation in patients with persistent atrium fibrillation.
Through incisions in the chest wall a thoracoscope is introduced to allow direct visualization of the heart.
Next the ablation device produces conduction blocks in areas like around the pulmonary veins to prevent erratic electric signals.
A clipping device called Atriclip is placed to close the left atrial appendage to avoid thrombi from this area (figure).
ICU monitoring device
.....
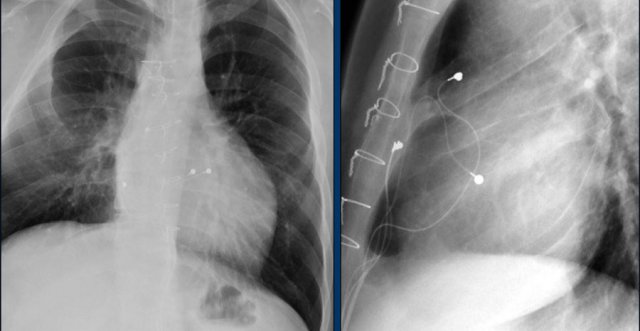
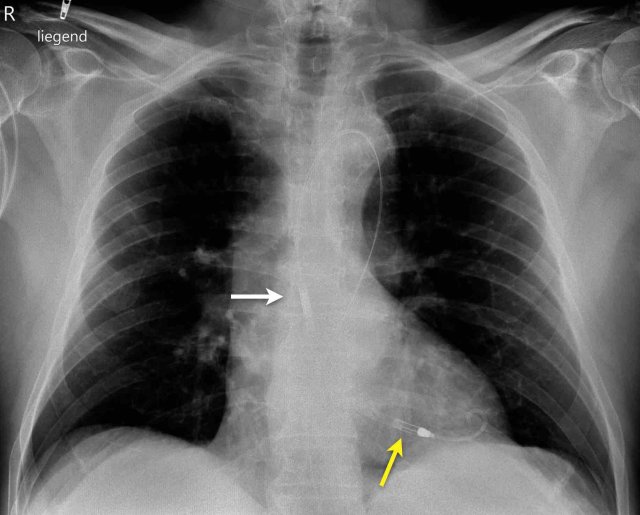
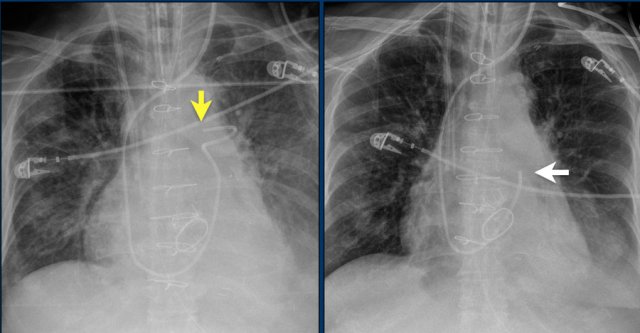
Swan Ganz catheter
The Swan GAnz catheter is a flow directed balloon tip catheter. The balloon is inflated within the left or right main pulmonary artery to measure the capillary wedge pressure and pulmonary artery pressure. Potential complications are intracardiac knotting, pulmonary infarction, pulmonary artery perforation, arrhythmias, cardiac perforation, and placement in the inferior vena cava.
The images show anincorrect placement of the Swan Ganz catheter on the left with the tip in the left upper lobe pulmonary artery branch.
The second X-ray shows adequate position of the catheter tip in the left pulmonary artery after repositioning.
Charity
All the profits of the Radiology Assistant go to Medical Action Myanmar which is run by Dr. Nini Tun and Dr. Frank Smithuis sr, who is a professor at Oxford university and happens to be the brother of Robin Smithuis.
Click here to watch the video of Medical Action Myanmar and if you like the Radiology Assistant, please support Medical Action Myanmar with a small gift.