Incidentalomas in the liver
What to do with incidentally found lesions in the liver?
Maarten van Leeuwen, Joost Nederend and Robin Smithuis
Radiology department of the University Medical Centre of Utrecht, the Leiden University and the Rijnland hospital, Leiderdorp, the Netherlands
Publicationdate
This review is based on a presentation given by Maarten van Leeuwen for the Dutch Radiology Society and was adapted for the Radiology Assistant by Joost Nederend and Robin Smithuis.
With the increasing use of multidetector CT small hepatic lesions are frequently depicted.
In many cases the pathological nature of these incidentally found liver lesions or incidentalomas is not known.
This results in a diagnostic problem, which is initiated by radiology so radiologists should take responsibility in correctly categorizing these lesions as to their clinical significance.
In this article we will discuss the management of two different type of incidentally found liver lesions:
- lesions that are too small to characterise (TSTC lesions) in asymptomatic individuals and in patients with a known malignancy.
- Hypervascular lesions.
TSTC (too small to characterize lesions)
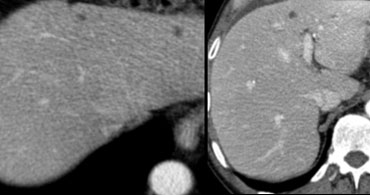
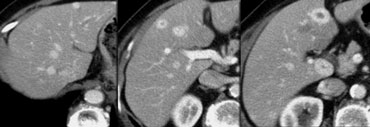
First study the images on the left.
Then continue reading.
We see multiple hypodense lesions.
We cannot diagnose them with certainty as:
- cysts: water-density, sharp demarcation
- hemangiomas: slowly progressive peripheral nodular enhancement of arterial density
- malignant lesions: inhomogeneous, irregular demarcation, peripheral enhancement less than arterial density
For this type of lesions which, due to their small size and atypical imaging features, cannot be confidently categorized, the term TSTC (to small to characterize) lesions has been coined.
TSTCs in patients without a known malignancy
Jones (1992) studied 1500 patients who had an abdominal CT examination (1).
He found:
- TSTC lesions in 17% of patients
- In 45 pts without a known malignancy, all lesions were benign
- In 209 pts with a known malignancy
- 86 had 1 TSTC: 5% malignant
- 74 had 2-4 TSTCs: 19% malignant
- 49 with >5 TSTCs: 76% malignant.
TSTCs in patients with a primary malignancy
Schwartz (1999) studied 2978 patient with a known malignancy (2).
He found TSTCs in 12% of patients with a known malignancy.
In 88% of patients the lesions were benign and in 12% they proved to be metastases (1.4% of all patients).
The percentage of malignancy depended much on the known primary tumor.
Most metastases were found in patients with breast cancer.
This is in accordance with the observation that breast metastases usually present as multiple small lesions, while liver metastases of colorectal cancer and lymphoma usually present as a solitary or a few larger masses.
Robinson (2003) studied various characteristics of TSTCs and their correlation with malignancy (3).
The lesions where classified by their behavior on follow up CT, as either stable or unstable.
Image features of stable (benign) lesions where small size and sharp edge.
Heterogeneity and soft tissue attenuation were associated with unstable behavior, but only seen in a small minority of cases.
TSTCs in breastcarcinoma
Krakora (2004) studied the prognostic importance of small hypoattenuating hepatic lesions seen at initial CT in patients with breast cancer, who did not have definite hepatic metastases at initial examination (4).
One or more small hypoattenuating hepatic lesions (TSTCs ) were seen in 54 of 153 patients (35%).
During a median follow-up of 584 days definite hepatic metastases developed in 43 of 153 patients (28%).
No difference was found in the chance for development of liver metastases in patients with or without TSTCs at initial CT.
Krakora concluded that in patients with breast cancer, who do not have definite hepatic metastases at presentation, there is no evidence that small hypoattenuating hepatic lesions seen at initial CT contribute to an increased risk of subsequently developing hepatic metastases.
Conclusion
In a patient without a known malignancy these small hypodense lesions, as a rule, should be considered as benign.
In a patient with a known malignancy a single TSTC lesion can also be assumed to be benign.
Even multiple TSTCs in these patients are mostly benign, especially when they are small, sharply defined and hypodens.
In these latter cases you should not be too defensive!
Don't dictate 'we can't rule out metastases'.
In patients with breastcancer and no known livermetastases at presentation, these TSTC lesions have no positive predictive value for the development of livermetastases in the long term.
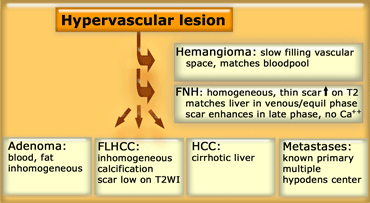
Incidental hypervascular lesions
Incidental hypervascular lesions are also very common findings in liver imaging.
It is important to differentiate between 'touch' and 'don't touch' lesions.
Benign 'don't touch' hypervascular tumors include hemangioma, FNH and small adenomas.
'Touch' lesions include large adenomas (more then 5 cm) and malignant tumors like Hepatocelular carcinoma (HCC), Fibrolamellar carcinoma (FLHCC) and metastases.
These enhancing, solid lesions should be differentiated from vascular lesions
, like hepatic aneurysm,
aortaportal shunt or pseudoaneurysm.
Incidence of hypervascular lesions
Karhunen (1986) found at autopsy an incidence of 20 % hemangioma, 3% FNH and 1% adenoma (5).
A study in 1989 by the AFIP showed a FNH : adenoma ratio of 8:1 in a series of 9000 autopsies (6).
Enhancement in Hemangioma
A hemangioma is a slowly perfused vascular space.
So the timing and amount of enhancement will
follow, but lag behind the arterial system.
Hemangiomas less than 1 cm frequently demonstrate
immediate homogenous enhancement, isodense to the aorta.
Hemangiomas larger than 1cm generally show slow
centripetal spread of nodular enhancement, slowly decreasing in density.
On the left a typical hemangioma.
Enhancement in arterial phase is almost isodense to the aorta, and, as contrast diffuses toward the center of the lesion, the level of enhancement lowers slowly, and in the late phase is still hyperdense compared to the vascular spaces.
 'Capillary blush' in FNH. Notice early enhancement, but not as bright as in hemangioma. In venous and delayed phase the enhancement is almost isodense to the liver.
'Capillary blush' in FNH. Notice early enhancement, but not as bright as in hemangioma. In venous and delayed phase the enhancement is almost isodense to the liver.
Enhancement in 'capillary blush'
The typical, slowly perfused vascular space enhancement of a hemangioma has
to be differentiated from the 'capillary blush' due to an abundant capillary network
which characterizes FNH, adenoma, HCC and
hypervascular metastases.
As capillaries are surrounded by tissue the overall enhancement will be less
dense compared to the
enhancement of the vascular spaces in
hemangioma.
Hence, in capillary blush, the enhancement occurs slightly later compared to the aorta and is less dense than the aorta.
Hemangioma
Hemangiomas on dynamic MR will show the same
enhancement characteristics as on contrast-enhanced CT.
The advantage of MR over CT is its higher sensitivity to contrast as will be shown in the next case.
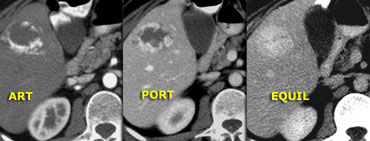
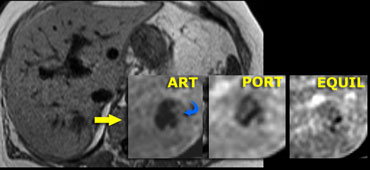
On the left an atypical, apparently hypovascular lesion on CT, possibly metastasis.
Same case on dynamic MR.
Notice how MR depicts the nodular, peripheral, slowly progressing enhancement (blue curved arrow) which CT failed to depict.
On the left an atypical hypoechoic lesion, surrounded by a small but definite halo.
In the arterial phase there is homogeneous
enhancement of arterial intensity, frequently seen in small hemangiomas.
In the portal venous phase and in the equilibrium phase it has the same enhancement as the aorta.
So all appearances are consistent with a hemangioma, a benign, non-solid
vascular lesion.
Once we have excluded hemangiomas, our
main goal is to determine whether a hypervascular lesion is a
FNH, which is the most prevalent hypervascular
solid lesion, or whether it is a lesion
which needs further management like adenoma,
HCC, FLHCC or hypervascular metastases.
For this purpose we have to look for morphologic features
like inhomogeneity and presence of capsule, scar,
calcification or fat.
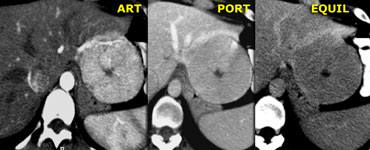
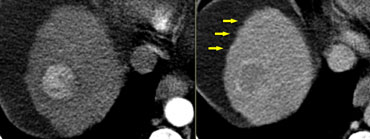
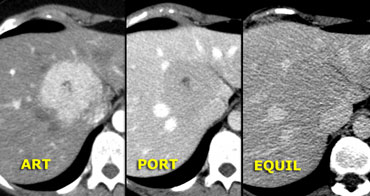
Focal Nodular Hyperplasia (FNH)
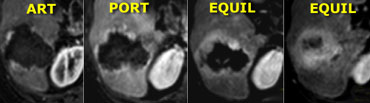
On the left two adjacent hypervascular lesions with
homogeneous enhancement in arterial phase and hypodense
central scars in arterial and venous phase, which
enhance in the equilibrium phase.
This is characteristic of FNH.
Notice that the small FNH, which is
anterior and right to the bigger one, has the same enhancement pattern.
FNH is considered a non-neoplastic, hyperplastic
response to a congenital vascular malformation.
Histologically, FNH is not a tumor and
consists of benign-appearing hepatocytes
occurring in a liver that is otherwise normal (i.e. no
cirrhosis).
At late arterial phase, FNH typically presents
with a bright homogeneous enhancement, but less intense than the aorta with
a hypodense central scar.
Smaller (
The radiating hypodense fibrous bands or
septa, arising from the scar, are not infrequent and
quite characteristic.
At portal phase, FNH is often iso-attenuating
to the normal liver and may be difficult to
deliniate.
Delayed phase often shows hyperattenuation of
the central scar and septa due to late
opacification of the fibrotic components.
No calcifications, inhomogeneity or capsule should be seen
in FNH.
Focal Nodular Hyperplasia (2)
On the left a typical FNH on MR.
Slightly hypointense on T1WI and slightly
hyperintense on T2WI.
The scar is somewhat hyperintense
on T2.
The enhancement is as we
expect with 'capillary blush' with a scar that
enhances late in the equilibrium phase.
Focal Nodular Hyperplasia (3)
On the left a lesion, that has all the
characteristics of FNH except for lack of late
enhancement of the central scar.
In addition, it is slightly hypodense to normal parenchyma in
the portal and equilibrium phase.
However, all other characteristics are present
like lobular enhancement, central scar and no
capsule, and therefore we characterize this lesion as FNH.
Focal Nodular Hyperplasia (4)
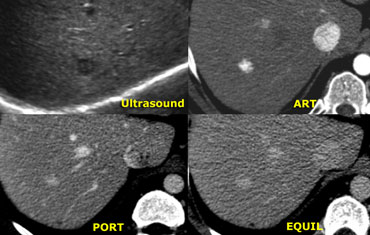
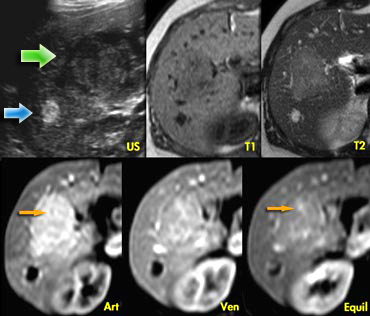
The ultrasound image on the left shows two lesions.
The small one (blue arrow) is characteristic of a
hemangioma, while the larger one (green arrow) is non
specific on US.
On T2WI the hemangioma shows the typical
homogeneous hyperintensity .
The larger lesion is somewhat hypointense on T1 and somewhat hyperintense on T2.
The enhancement is almost homogeneous with
small septae that do not enhance in the arterial
phase and do show late enhancement (yellow arrows).
On T2WI the hemangioma shows the typical
homogeneous hyperintensity .
The larger lesion is somewhat hypointense on T1 and somewhat hyperintense on T2.
The enhancement is almost homogeneous with
small septae that do not enhance in the arterial
phase, and do show late enhancement (yellow arrows).
We also characterize this lesion as FNH.
Focal Nodular Hyperplasia (5)
On the left another FNH on MR.
The lesion is almost isointense to liver on T1WI and T2WI, but shows more contrast to the liver on a T1W-MPRGRE (gradient-echo).
The enhancement in the arterial phase is lobulated with nonenhancing septation and in the equilibrium phase the lesion is not different from normal liver parenchyma.
Notice that the lesion has a small scar.
Small FNHs often do not have a central scar on imaging and even not on pathologic examination.
Focal Nodular Hyperplasia (6)
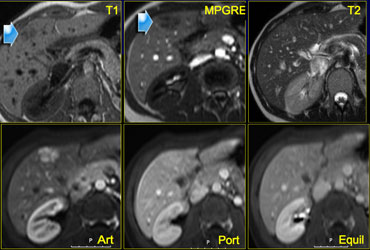
Another FNH on the left, in order to get really familiar with these common lesions.
On a CTA for pulmonary emboli a small hypervascular lesion is seen in the liver.
Further evaluation was done with MR.
On T1WI the lesion is not seen and on T2WI it is only slightly hyperintense.
In the arterial phase there is homogeneous enhancement and in the venous phase the lesion is not seen.
Provided that this patient does not have liver cirrhosis, this is probably a benign lesion, probably FNH.
As the appearance was not pathognomonic for FNH, a follow up examination was done and the lesion had not changed, making the
diagnosis FNH most likely.
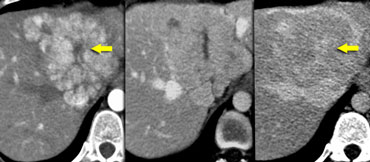
When does it stop, this comfortable feeling, that something is a FNH?
It stops when there are too many features that do not belong to a FNH.
Like the case on the left.
Decide for yourself which findings are compatible with the diagnosis typical FNH and which are not.
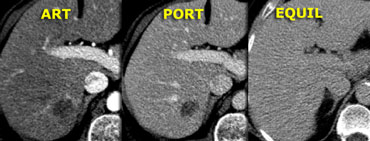
The lesion on the left does have a central scar
like FNH , but on the T1WI the lesion is inhomogeneous and not sharply defined.
On T2WI the scar has a low signal intensity.
For typical FNH the signal intensity however should be high and the lesion is again
inhomogeneous.
In the arterial phase the lesion does enhance
like FNH, but in the portal and equilibrium
phase the enhancement persists and is inhomogeneous.
In addition, the central scar does not enhance in the
late phase.
So there are many findings that are not
compatible with the diagnosis FNH.
Since the specificity for diagnosing a lesion as
benign should be very high, we cannot stop
here and we have to get a histological diagnosis.
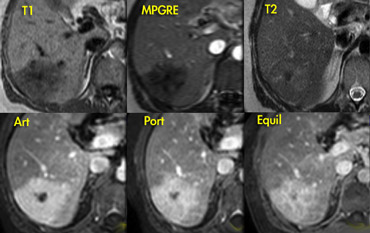
Fibrolamellar HCC
When we encounter lobulated hypervascular masses in the liver, an important diagnosis that you don't want to miss is a fibrolamellar hepatocellular carcinoma (FLHCC).
This particular form of HCC may mimick FNH on imaging.
In contrast to HCC, the prognosis is reasonable.
Like FNH, FLHCC also is a hypervascular, lobulated mass with a central scar
.
Both FNH and FLHCC appear in normal liver, unlike
HCC that is most frequently seen in a cirrhotic liver.
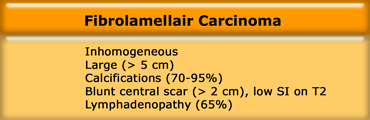
In distinction to FNH, FLHCC is inhomogeneous,
large (> 5 cm), frequently has calcifications (>70%), a
blunt central scar and usually there is
lymphadenopathy.
Calcifications in FNH are so uncommon that it
should make you consider another diagnosis like
FLHCC.
Fibrolamellar HCC (2)
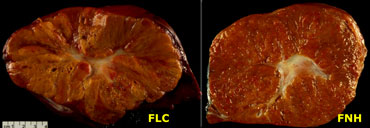
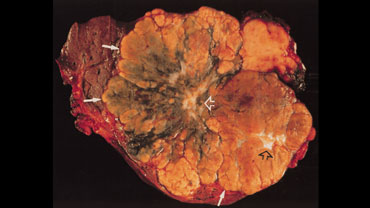
On the left a pathologic specimen of FLHCC and FNH.
At first glance they look very similar.
However when you look carefully you will notice the more lamellar and heterogeneous structure of FLHCC compared to the homogeneous appearance of FNH.
Fibrolamellar HCC (3)
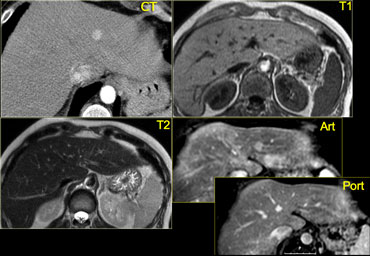
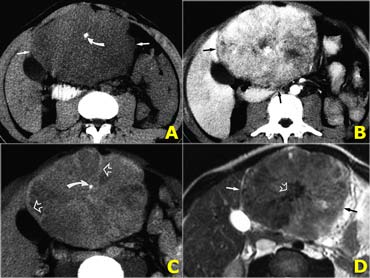
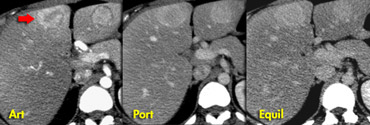
On the left CT- and MR-images of a left-lobe fibrolamellar HCC in a 19-year-old man.
A. Non-enhanced transverse CT scan shows calcification (curved arrow) within the hypoattenuating tumor (straight arrows).
B. Hepatic arterial contrast-enhanced transverse CT scan shows heterogeneous hypervascularity within the tumor (arrows).
C. Ten-minute delayed transverse CT scan demonstrates subtle areas of hyperattenuation that represent fibrous tissue within the central scar, radiating septa, and capsule (open arrows).
Curved arrow = calcification.
D. Transverse T2-weighted MR image (5,000/105) also demonstrates the central scar and septa (open arrow).
The tumor itself (straight arrows) is nearly isointense to liver (the only such case in our series).
On the left a photograph of the cut surface of the gross pathologic specimen shows a large tumor with eccentric and central scars (open arrows) and radiating septa.
The mass has an irregular lobulated pushing margin (solid arrows) and a variegated appearance with areas of bile staining.
In a series of 31 cases of FLHCC, Ichikawa et al (7) found the following:
- Large tumors (mean diameter, 13 cm) were depicted at CT and MR in all cases.
- At CT, the margins of the tumors were well defined in 24 (77%) of 31 cases.
- Calcifications were depicted in 21 (68%)
- Central scar in 22 (71%)
- Abdominal lymphadenopathy in 20 (65%).
- In 20 (80%) of 25 cases with hepatic arterial phase CT images, tumors were heterogeneous and depicted areas of hypervascularity.
- At MR imaging, tumors were hypointense to liver on TI-weighted images (n = 11) and hyperintense to liver on T2-weighted images (n = 10).
- Calcification was not depicted on MR images, but a central scar was depicted as hypointense to surrounding tumor in nine cases.
Adenoma
An adenoma is regularly characterized by bleeding, fat or peliosis.
Although we cannot see peliosis itself, it can result in a hyperintense lesion on T1WI.
On the left two incidentalomas.
Decide for yourself why these are not FNH lesions.
In the arterial phase there are two
hypervascular lesions, somewhat less
dense than we would expect in FNH.
Both lesions demonstrate a halo of a capsule,
which should not be apparent in FNH.
Unlike in FNH, the enhancement is
inhomogeneous and in the portovenous and
equilibrium phase the lesions are not isodens to
the liver.
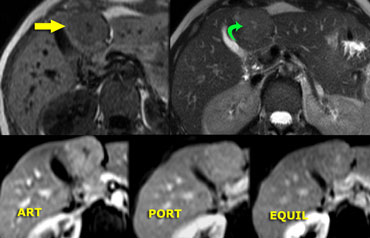
Adenoma (2)
Regularly adenomas present with bleeding.
On the left images of a woman who presented with acute abdominal pain.
On US a livermass was seen and free fluid surrounding the liver.
This is a typical presentation of an adenoma.
On portal phase CT, the lesion is hypointense with haemorrhage adjacent to the lesion, extending subcapsularly.
Adenoma (3)
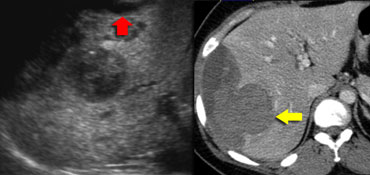
On the left an US image of an incidentally found lesion in a 50 y old female.
Work up was done with CT, but only non-specific features were found without signs of hypervascularity. .
Continue with next images.
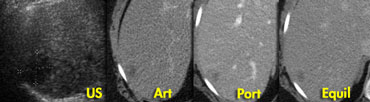
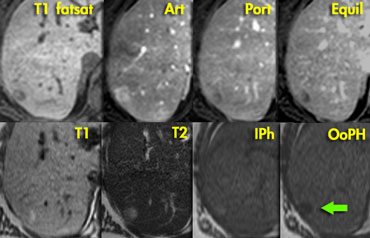 Adenoma: capillary blush in arterial phase and signal loss in out of phase image indicating the presence of fat.
Adenoma: capillary blush in arterial phase and signal loss in out of phase image indicating the presence of fat.
In contrast to the CT, there clearly is
enhancement in the arterial phase on MR, again demonstrating that MR
depicts enhancement better than CT.
The enhancement is due to a capillary blush, most intense in the arterial phase with apparent wash-out in portal and equilibrium phase, due to greater enhancement of the surrounding parenchyma.
In the 'out of phase' image there is signal loss
indicating that the lesion contains fat,
which is very suggestive for adenoma.
A HCC may also contain fat, but in this case
there is no cirrhosis and the entire
lesion shows signal loss,
which we would not expect in HCC.
Hepatocellular carcinoma (HCC)
Concerning the diagnosis of HCC, there is
one thing to remember: 'Every hypervascular lesion in a cirrhotic liver is
HCC until proven otherwise'
'.
On the left we see a cirrhotic liver with irregular
margins (arrows), suggesting that the hypervascular lesion is a HCC.
The inhomogeneous
enhancement and the partial capsule are helpful
for the diagnosis HCC, but even if these
features were not present, our diagnosis still
would be HCC.
Hypervascular metastases
Characteristics of hypervascular metastases are:
- A hypervascular primary tumor like endocrine tumors (thyroid, carcinoid), renal cell tumors and some breast carcinomas.
- Often coexisting hypo- and hypervascular metastases.
- Larger lesions are often inhomogeneous due to central necrosis.
On the left hypervascular metastases.
Notice that the larger ones show central necrosis, as they outgrow their blood supply.
Differential diagnosis of Hypervascular lesions
Work up
In the workup of incidentally found
hypervascular lesions, we first have to decide
whether the lesion is a hemangioma, because
these are the most common lesions and usually have
specific imaging findings.
If not, we have to find out whether it is an FNH.
For this differentiation we have to look at
differences in enhancement pattern and
differences in morphology like presence of a
capsule, scar, calcification and inhomogeneity.
Hypervascular lesions most often can be characterized, even when small.
FNH and hemangiomas need no further investigation or treatment.
The preferred modality to characterize incidentalomas is MR, as it is better for lesion characterization and incidentalomas often occur in young females, where radiation burden should be minimized.
If HCC or FLHCC is considered further investigation is always needed.
Differential diagnosis
In the table on the left we have summarized the typical findings in FNH, Adenoma and HCC.
Since FNH is so common, we have to get a clear mental picture of the many ways that these lesions present.
As radiologists we have a great responsibility here.
In FNH not all features have to be present, but there should be no calcification or high signal intensity on T1WI and the lesion should not be inhomogeneous or have a capsule.
Sometimes the term 'stealth lesion' is used to describe the phenomenon that some of these small FNH lesions are only seen in the arterial phase.
Charity
All the profits of the Radiology Assistant go to Medical Action Myanmar which is run by Dr. Nini Tun and Dr. Frank Smithuis sr, who is a professor at Oxford university and happens to be the brother of Robin Smithuis.
Click here to watch the video of Medical Action Myanmar and if you like the Radiology Assistant, please support Medical Action Myanmar with a small gift.