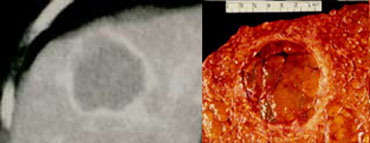
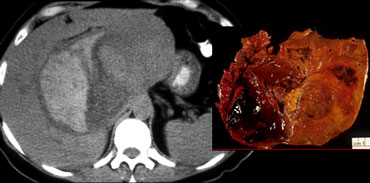
Characterisation of liver masses
Richard Baron
Radiology department of the University of Chicago
Publicationdate
This article is based on a presentation given by Richard Baron and adapted for the Radiology Assistant by Robin Smithuis.
Richard Baron is Chair of Radiology at the University of Chicago and well known for his work on hepatobiliary diseases.
He has been president of the Society of Computed Body Tomography and Magnetic Resonance.
In Part I a basic concept is given on how to detect and characterize livermasses with CT.
In Part II the imaging features of the most common hepatic tumors are presented.
Interactive cases are presented in the menubar to test your knowledge (Liver mass 1 and 2).
Detection of liver masses
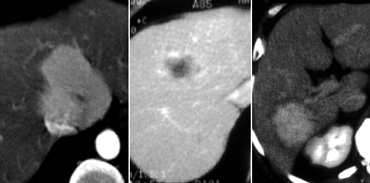
 Small Hepatocellular carcinoma in cirrhotic liver not visible on NECT (left), clearly visible in arterial phase (middle) and not visible in portal venous phase (right)
Small Hepatocellular carcinoma in cirrhotic liver not visible on NECT (left), clearly visible in arterial phase (middle) and not visible in portal venous phase (right)
The conspicuity of a liver lesion depends on the attenuation difference between the lesion and the normal liver.
On a non enhanced CT-scan (NECT) liver tumors usually are not visible, because the inherent contrast between tumor tissue and the surrounding liver parenchyma is too low.
Only a minority of tumors contain calcifications, cystic components, fat or hemorrage and will be detected on a NECT.
So i.v. contrast is needed to increase the conspicuity of lesions.
When we give i.v. contrast, it is important to understand, that there is a dual blood supply to the liver.
Normal parenchyma is supplied for 80% by the portal vein and only for 20% by the hepatic artery, so it will enhance in the portal venous phase.
All liver tumors however get 100% of their blood supply from the hepatic artery, so when they enhance it will be in the arterial phase.
This difference in bloodsupply results in different enhancement patterns between liver tumors and normal liver parenchyma in the various phases of contrast enhancement (figure).
 Detection of a lesion depends on difference in attenuation between liver and lesion.LEFT: Arterial phase showing hypervascular FNHMIDDLE: Portal venous phase showing hypovascular metastasisRIGHT: equilibrium phase showing relatively dense cholangiocarcinoma
Detection of a lesion depends on difference in attenuation between liver and lesion.LEFT: Arterial phase showing hypervascular FNHMIDDLE: Portal venous phase showing hypovascular metastasisRIGHT: equilibrium phase showing relatively dense cholangiocarcinoma
In the arterial phase hypervascular tumors will enhance via the hepatic artery, when normal liver parenchyma does not yet enhances, because contrast is not yet in the portal venous system.
These hypervascular tumors will be visible as hyperdense lesions in a relatively hypodense liver.
However when the surrounding liver parenchyma starts to enhance in the portal venous phase, these hypervascular lesion may become obscured.
In the portal venous phase hypovascular tumors are detected, when the normal liver parenchyma enhances maximally.
These hypovascular tumors will be visible as hypodense lesions in a relatively hyperdense liver.
In the equilibrium phase at about 10 minutes after contrast injection, tumors become visible, that either loose their contrast slower than normal liver, or wash out their contrast faster than normal liver parenchyma.
These lesions will become either relatively hyperdense or hypodense to the normal liver.
Arterial phase imaging
Optimal timing and speed of contrast injection are very important for good arterial phase imaging.
Hypervascular tumors will enhance optimally at 35 sec after contrast injection (late arterial phase).
This time is needed for the contrast to get from the peripheral vein to the hepatic artery and to diffuse into the liver tumor.
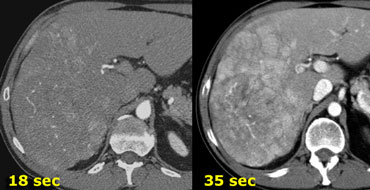
On the left a patient who underwent two phases of arterial imaging at 18 and 35 seconds.
In the early arterial phase we nicely see the arteries, but we only see some irregular enhancement within the liver.
In the late arterial phase we can clearly identify multiple tumor masses.
Notice that in the late arterial phase there has to be some enhancement of the portal vein.
The only time that an early arterial phase is needed is when you need an arteriogram, for instance as a roadmap for chemoembolization of a liver tumor.
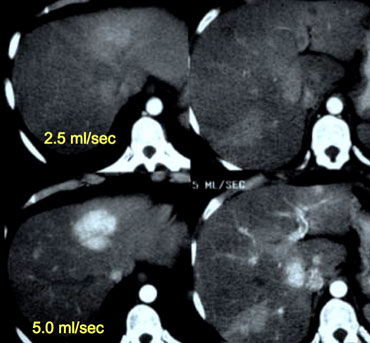 Patient with liver cirrhosis and multifocal HCC injected at 2.5ml/sec (left) and at 5ml/sec (right).
Patient with liver cirrhosis and multifocal HCC injected at 2.5ml/sec (left) and at 5ml/sec (right).
Timing of scanning is important, but almost as important is speed of contrast injection.
For arterial phase imaging the best results are with an injection rate of 5ml/sec.
There are two reasons for this better enhancement: at 5ml/sec there will be more contrast delivered to the liver when you start scanning and this contrast arrives in a higher concentration.
On the left a patient with cirrhosis examined after contrast injection at 2.5ml/sec and at 5ml/sec.
At 5ml/sec there is far better contrast enhancement and better tumor detection.
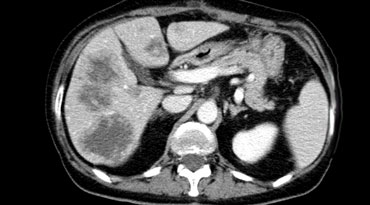 Hypovascular metastases seen as hypodense lesions in the late portal venous phase. Notice some rim enhancement of the more viable peripheral areas of the metastases.
Hypovascular metastases seen as hypodense lesions in the late portal venous phase. Notice some rim enhancement of the more viable peripheral areas of the metastases.
Portal Venous phase
Portal venous phase imaging works on the opposite idea.
We image the liver when it is loaded with contrast through the portal vein to detect hypovascular tumors (figure).
The best moment to start scanning is at about 75 seconds, so this is a late portal venous phase, because enhancement of the portal vein already starts at 35 sec in the late arterial phase.
This late portal venous phase is also called the hepatic phase because there already must be enhancement of the hepatic veins.
If you do not seen enhancement of the hepatic veins, you are too early.
If you only do portal venous imaging, for instance if you are only looking for hypovascular metastases in colorectal cancer, fast contrast injection is not needed, because in this phase the total amount of contrast is more important and 3ml/sec will be sufficient.
Equilibrium Phase
The equilibrium phase is when contrast is moving away from the liver and the liver starts to decrease in density.
This phase begins at about 3-4 minutes after contrast injection and imaging is best done at 10 minutes after contrast injection.
This phase can be valuable if you're looking for: fast tumor washout in hypervascular tumors like HCC or retention of contrast in the blood pool as in hemangiomas or the retention of contrast in fibrous tissue in capsules (HCC) or scar tissue (FNH, Cholangioca).
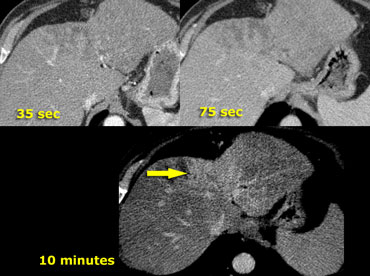
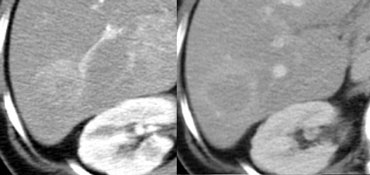
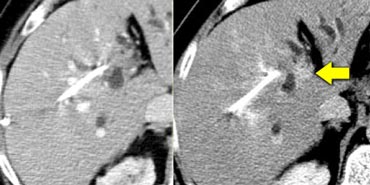 Small cholangiocarcinoma not visible in portal venous phase (left), but seen as relative hyperdense lesion in the delayed phase (right).
Small cholangiocarcinoma not visible in portal venous phase (left), but seen as relative hyperdense lesion in the delayed phase (right).
Relative hyperdense lesions in the delayed phase
Fibrous tissue that's well organized and dense is very slow to let iodine or gadolineum in.
Once contrast gets in however, it is equally slow to get back out in the equilibrium phase.
So when the normal liver parenchyma washes out, the fibrous components of a tumor will look brighter than the background liver tissue.
Cholangiocarcinoma may have a fibrous stroma and in the delayed phase it may be the only time when you see the tumor (figure).
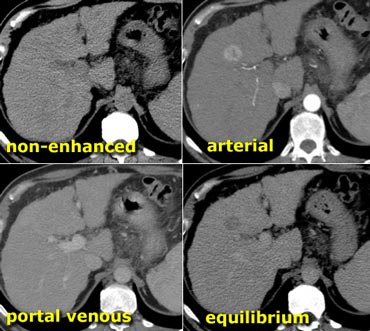 HCC in a cirrhotic liver. Notice fast wash out in equilibrium phase compared to surrounding liver parenchyma.
HCC in a cirrhotic liver. Notice fast wash out in equilibrium phase compared to surrounding liver parenchyma.
Relative hypodense lesions in the delayed phase
On the left the importance of the delayed phase in a cirrhotic patient with an HCC is demonstrated.
Notice that you do not see the tumor on the nonenhanced scan and also not in the portal venous phase. This is often the case and demonstrates the importance of the arterial phase.
Now the issue at hand is in small enhancing lesions in a cirrhotic liver whether it is a benign lesion like a regenerating nodule or a HCC.
In the delayed phase we see that the tumor is washed out more than the surrounding liver parenchyma.
Benign lesions typically will not show this kind of wash out.
For instance a FNH or adenoma will show fast enhancement in the arterial phase, become isodense in the portal venous phase, but it will stay isodense with liver in the equilibrium phase.
These benign tumors do not have enough neoplastic neovascularity to have a fast wash out.
Especially in cirrhotic patients you have to rely heavily on this delayed phase to differentiate benign little enhancing lesions from small HCC's.
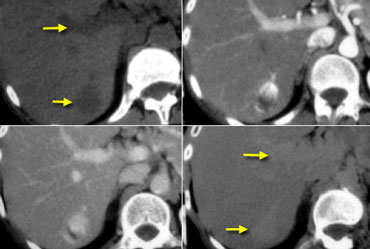 Hemangioma on NECT, late arterial, late portal venous and equilibrium phase. Notice that the attenuation of the hemangioma matches the bloodpool in every single phase (arrows).
Hemangioma on NECT, late arterial, late portal venous and equilibrium phase. Notice that the attenuation of the hemangioma matches the bloodpool in every single phase (arrows).
Blood pool and Hemangioma
Normally when we look at lesions filling with contrast, the density of these lesions is always compared to the density of the liver parenchyma.
In hemangiomas however you should not compare the density of the lesion to the liver, but to the blood pool.
This means that the areas of enhancement in a hemangioma should match the attenuation of the appropriate vessels (bloodpool) at all times.
So in the arterial phase the enhancing parts of the lesion must have almost the same attenuation value as the enhancing aorta , while in the portal venous phase it must match the enhancement of the portal vein.
If it does not match the bloodpool in every single phase of contrast enhancement forget the diagnosis of a hemangioma.
On the left a characteristic hemangioma.
Notice that on the NECT the density of the tumor is the same as the density of the vessels.
In the arterial phase it is matching the bloodpool and the attenuation is almost the same as the aorta.
In the portal venous phase it matches the density of the portal vein.
In the equlibrium phase it has the same enhancement as the vessels.
Eventually the lesion will become iso-attenuating to the liver, but only because the vessels become iso-attenuating with the liver.
It has nothing to do with the density of the liver parenchyma itself.
So think of bloodpool rather than liver if you're thinking of a hemangioma.
Tailored CT protocol
You have to adapt your protocol to the type of scanner, the speed of contrast injection and to the kind of patient that you are examining.
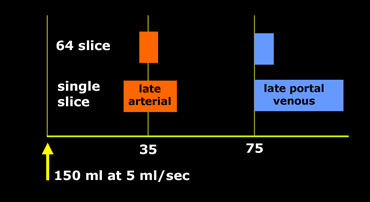
If you have a single slice scanner, it will take about 20 seconds to scan the liver.
For late arterial phase imaging 35 sec is the optimal time, so you start at about 25 seconds and end at about 45 seconds.
However if you have a 64-slice scanner, you will be able to examine the whole liver in 4 seconds. So you start scanning at about 33 seconds, which is much later.
In aterial phase imaging the time window is narrow, since you have only limited time before the surrounding liver will start to enhance and obscure a hypervascular lesion.
For portal venous phase imaging it is different. Here you don't want to be too early, because you want to load the liver with contrast and it takes time for contrast to get from the portal vein into the liver parenchyma.
Besides you have more time, because the delayed or equilibrium phase starts at about 3-4 minutes.
So you start at 75 seconds with whatever scanner you have.
Only when you inject with high speed at 5ml/sec you may start earlier at about 65-70 seconds.
Use arterial phase imaging in the following situations:
- Characterisaton of a liver lesion of unknown origin.
- Detection of HCC in patients with a high alpha 1 foetoprotein.
- Screening of cirrhotic patients for HCC.
- Detection of metastases in patients with hypervascular tumors.
Characterisation of liver masses
From a practical point of view, the approach to characterizing a focal liver lesion seen on CT begins with the determination of its density.
If the lesion is of near water density, homogeneous, has sharp margins and shows no enhancement, then it is a cyst.
If the lesion does enhance, then the next step is to determine whether the lesion could be a hemangioma, since this is by far the most common liver tumor. The enhancement should be peripheral and nodular, with the same density as the bloodpool in all phases.
If it is not a cyst nor a hemangioma, then we further have to study the lesion.
Based on the enhancement pattern, we divide masses into hypervascular and hypovascular lesions.
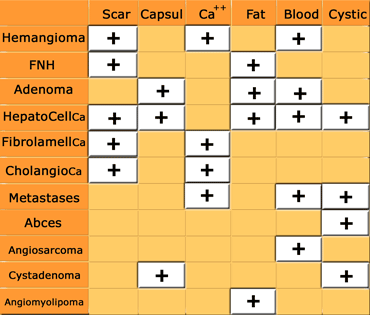
Usually a combination of the enhancement pattern and gross pathologic features, like the presence of fat, blood, calcifications, cystic or fibrotic components, in combination with the clinical history will limit the differential diagnosis (figure).
Hypervascular lesions
Arterially enhancing lesions are mostly benign lesions and include primary liver tumors as FNH, adenoma and small hemangiomas that fill rapidly with contrast.
These benign tumors have to be differentiated from the most common hypervascular malignant liver tumor, which is HCC and metastases from hypervascular tumors like melanoma, renal cell carcinoma, breast, sarcoma and neuroendocrine tumors (islet cell tumors, carcinoid, pheochromocytoma).
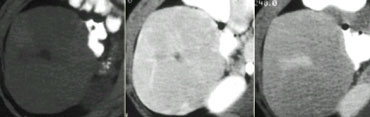
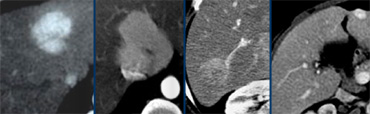 Four different tumors with enhancement in the late arterial phase. From left to right: HCC in cirrhotic liver; FNH with central scar in adolescent; adenoma in young woman on contraceptives and finally a hemangioma with typical enhancement in the other phases (not shown).
Four different tumors with enhancement in the late arterial phase. From left to right: HCC in cirrhotic liver; FNH with central scar in adolescent; adenoma in young woman on contraceptives and finally a hemangioma with typical enhancement in the other phases (not shown).
Hypervascular lesions may look very similar in the arterial phase (figure).
Differentiation is done by looking at the enhancement pattern in the other phases and additional gross pathologic features together with clinical findings.
Hypervascular metastases will be considered in patients with a known primary tumor.
In general HCC is considered when there is a setting of cirrhosis, while FNH is considered in young women and hepatic adenoma in patients on oral contraceptives, anabolic steroids or with a history of glycogen storage disease.
Hypovascular lesions
Hypovascular liver tumors are more common than hypervascular tumors.
Most hypovascular lesions are malignant and metastases are by far the most common.
Although primary liver tumors are mostly hypervascular, there are exceptions. 10% of HCC is hypovascular. Cholangioca is hypovascular, but may show delayed enhancement (figure).
On the left a hypovascular mass with irregular enhancement in the late arterial and late portal venous phase. This is a sign of malignancy.
On the delayed images a relative dense structure is seen centrally, which looses its contrast slower compared to normal liver.
This means that this tumor is mainly composed of fibrous tissue.
The fibrous tissue has also retracted the liver capsule.
These imaging findings are very suggestive of a cholangiocarcinoma.
Scar
Liver lesions which may have a central scar are FNH, fibrolamellar carcinoma, cholangiocarcinoma, hemangioma and hepatocellular carcinoma.
On CT a scar is sometimes visible as a hypodense structure.
On MR scar tissue is hypointense on both T1WI and T2WI due to intense fibrotic changes. An example is the central scar of fibrolamellar carcinoma (FLC)
An exception to this rule is the central scar in FNH which is hyperintense on T2WI due to edema.
T2WI can be very helpfull if there is a problem in differentiating FNH from FLC.
Both on CT and MRI scar tissue will enhance in the delayed phase.
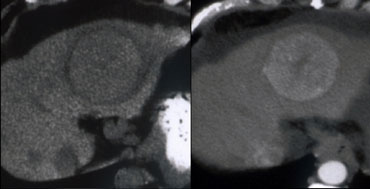
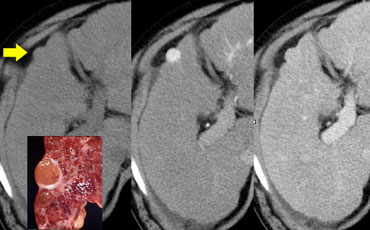
On the left a lesion with a typical central scar. It has a hypodense centre on the NECT.
In the portal venous phase there is homogeneus enhancement of the lesion except for the scar.
Enhancement of the fibrous tissue of the central scar is seen only on the delayed phase images.
The combination of homogeneous enhancement and central scar is typical for the diagnosis of FNH.
Capsule
Liver lesions which may have a capsule are Adenoma, HCC and cystadenoma or cystadenocarcinoma.
The most common tumor with a capsule is HCC.
The capsule will not enhance in the arterial phase and even in the portal venous phase it will be hypodense, because the fibrous tissue enhances very slowly.
A capsule is usually best seen in the delayed phase as a relative hyperdense structure.
Adenoma frequently has a thin fibrous capsule seen in 30% of cases.
It has a well defined contour and subcapsular feeding arteries.
On the left a different patient with HCC.
Only in the equilibrium phase a relatively bright capsule was seen.
The image on the left was taken 8 minutes after contrast injection.
Notice that the tumor itself is relative hypodense in the equilibrium phase.
So it has a fast wash out.
Calcifications
Central calcifications are seen in:
- Metastases (especially in colorectal tumors)
- Fibrolamellar carcinoma (FLC)
- Cholangiocarcinomas
- Hemangiomas
These calcifications are hyperdense on CT and hypointense on T1 and T2 MR images.
In FLC these calcifications are located within the central scar as seen on the left.
Fat
Fat within liver tumors is seen in:
- Adenoma
- HCC
- Metastatic liposarcoma
- Angiomyolipoma
The case on the left shows an adenoma with fat depositions within the tumor.
Hemorrhage
Hemorrhage in liver tumors is seen in:
- Adenoma
- HCC
Hemorrhage is most commonly seen in adenomas.
The case on the left shows a well circumscribed lesion with hemorrhage.
At resection the lesion proved to be an adenoma.
Cystic components
If a lesion has a near water density in the centre and does not show enhancement in the centre, we usually will call it a cystic lesion.
You have to realize, that it still can be a tumor as in cystic metastases or metastases with central necrosis.
Secondly you always have to add absces to the differential diagnosis.
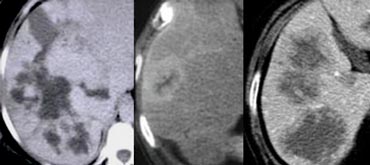
On the left a patient with hypovascular lesions with a low density, so it may be cystic i.e fluid containing.
These lesions are multiple, but not spread out through the liver, so we describe them as clustered or satelite lesions.
This is a typical finding which makes the lesions suspective for liver abcesses.
This was a case of diverticulitis. The common route is through the portal vein as a result of abdominal infection. The bacteria enter the slow flow portal system, where they layer within the vessel and finally these bacteria 'fall down' into the dependent portion of the right lobe.
On the left a typical case of a echinococcus cyst with 'daughter cysts' within the large cyst.
Most cases of echinococcus cysts however are not that typical.
If you look at the CT image on the left, the first impression might be that there are only simple cysts within the liver.
However, if you look more carefully, you will notice that some of the hypodense lesions show vague rim enhancement.
And although you might think that these could be cystic metastases, the US-findings clearly show, that these lesions are hyperechoic solid masses.
So you have to be very carefull in calling a lesion cystic, because you might end up missing metastases or looking in the wrong file for a differential diagnosis.
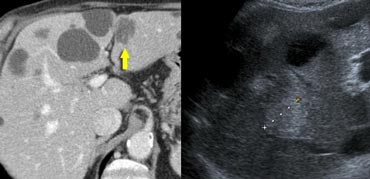
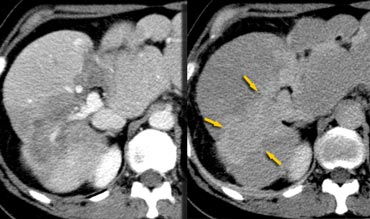 Hepatic and delayed phase in a patient with breast metastases causing retraction of liver capsule. Notice delayed enhancement of the fibrotic tissue (arrows)
Hepatic and delayed phase in a patient with breast metastases causing retraction of liver capsule. Notice delayed enhancement of the fibrotic tissue (arrows)
Retraction of liver capsule
Most liver tumors will present as a mass.
Some tumors however have an infiltrative growth pattern with a lot of fibrous tissue and do not cause mass effect.
As the fibrous stroma matures, the tissue will contract and cause retraction of the liver capsule (figure).
Breast cancer metastases can be infiltrative. When they shrink they can cause multiple retractions.
This will give a pseudo-cirrhosis appearance.
 Delayed phase image of a cholangiocarcinoma with relative dense fibrous stroma with retraction of liver capsule
Delayed phase image of a cholangiocarcinoma with relative dense fibrous stroma with retraction of liver capsule
The most common tumor however to cause retraction is cholangiocarcinoma.
The delayed image on the left shows a large cholangiocarcinoma with dense enhancing fibrous tissue and retraction of the liver capsule.
Notice the resemblance with the case above.
Another cause of local retraction is atrophy due to biliary obstruction or chronic portal venous obstruction.
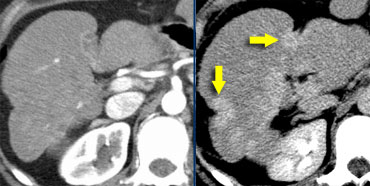 Hepatic and delayed phase in a patient with multifocal cholangiocarcinoma causing retraction of liver capsule.
Hepatic and delayed phase in a patient with multifocal cholangiocarcinoma causing retraction of liver capsule.
On the left another case of cholangiocarcinoma with multifocal lesions.
Notice the retraction and the delayed enhancement of the fibrotic component of the tumor.
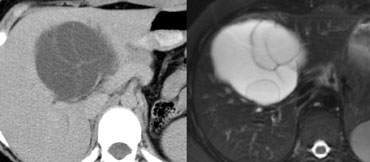
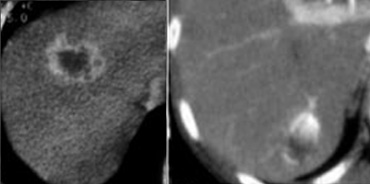 LEFT: rim enhancement in metastasis. RIGHT: hemangioma with discontinuous nodular peripheral enhancement
LEFT: rim enhancement in metastasis. RIGHT: hemangioma with discontinuous nodular peripheral enhancement
Peripheral enhancement and progressive fill in
Many will regard 'peripheral enhancement and progressive fill in' as a typical feature of hemangioma, but it is not.
Peripheral rim enhancement is a typical feature of malignant lesions and only discontinuous nodular peripheral enhancement that matches bloodpool is a typical feature of hemangioma.
Many lesions will show progressive fill in.
In hemangiomas this progressive fill in must have the same density as the bloodpool.
Many hypovascular metastases will show contrast diffusion into a lesion starting on the outside. Usually the center does not fill in.
Cholangiocarcinomas will show progressive fill in because the fibrous centre will enhance slowly. You will see it enhance in the delayed phase (see part II)
So if you want to make the diagnosis of a hemangioma you have to look at all the other phases to see if the enhancement matches the bloodpool.