Multiple Sclerosis 2.0
Diagnosis and differential diagnosis
Frederik Barkhof and Robin Smithuis
Amsterdam University Medical Center and University College London and Alrijne Hospital Leiderdorp, the Netherlands
Publicationdate
This article is an updated version of the 2013 article and focusses on the role of MRI in the diagnosis of Multiple Sclerosis.
We will discuss the following subjects:
- Typical findings in MS
- Role of MR in the McDonald criteria of MS
- How to differentiate MS lesions from other white matter diseases
- The importance of the a priori chance for the differential diagnosis of white matter lesions.
There is an important role for MRI in the diagnosis of MS, since MRI can show multiple lesions - dissemination in space, many of which are clinically occult already at the time of first presentation, and MRI can show new lesions on follow up scans - dissemination in time, much earlier than new symptoms develop.
Introduction
 * In elderly patients or patients with cardiovascular risk factors it is better to look for at least 3 periventricular lesions
* In elderly patients or patients with cardiovascular risk factors it is better to look for at least 3 periventricular lesions
McDonald criteria
More information:
Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria
Multiple sclerosis is the most common inflammatory demyelinating disease of the central nervous system in young and middle-age adults, but may also affect older people.
According to the McDonald criteria for MS, the diagnosis requires objective evidence of lesions disseminated in time and space, either clinically or radiologically and elimination of more likely diagnoses.
There is an important role for MRI in the diagnosis of MS, since MRI can show multiple lesions (dissemination in space), many of which are clinically occult already at the time of first presentation, and MRI can show new lesions on follow up scans (dissemination in time) much earlier than new symptoms develop.
The McDonald criteria are very specific, because if you want to use MRI for the diagnosis of MS, you must confrim that the patient really has MS.
You do not want a patient to start treatment daily if there is any doubt about the diagnosis.
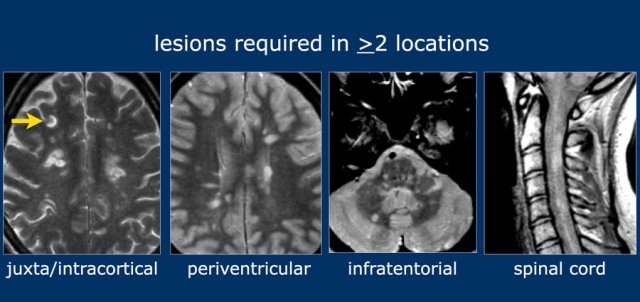
Dissemination in Space
For dissemination in space (DIS) lesions in two out of four typical areas of the CNS are required:
- juxtacortical/intracortical
- periventricular
- infratentorial
- spinal cord
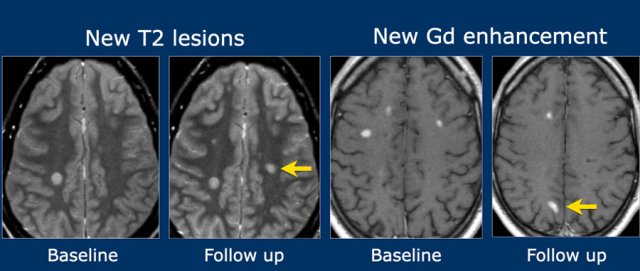
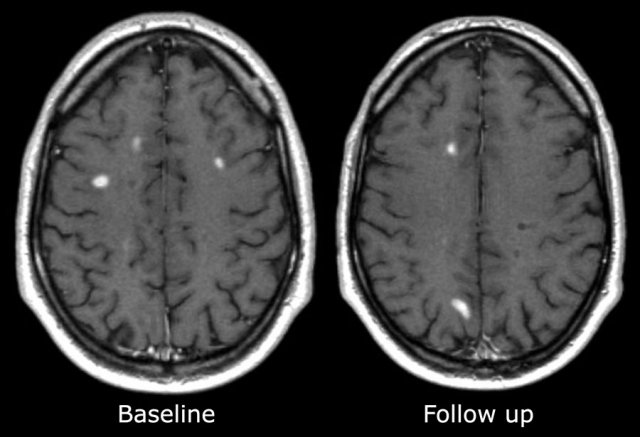
Dissemination in time
For dissemination in time (DIT) there are two possibilities:
- A new T2 and/or gadolinium-enhancing lesion(s) on follow-up MRI, with reference to a baseline scan, irrespective of the timing of the baseline MRI OR
- Simultaneous presence of asymptomatic gadolinium-enhancing and non-enhancing lesions at any time.
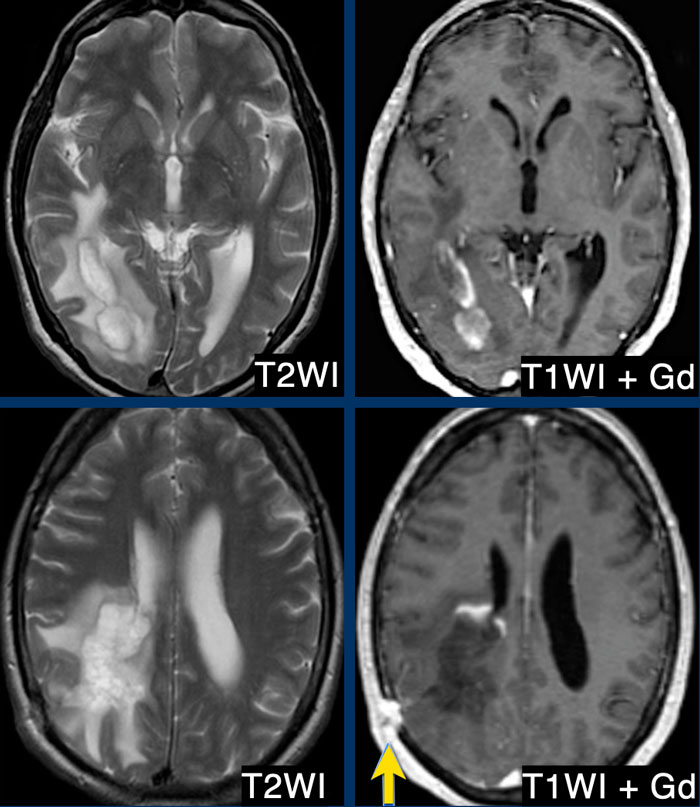
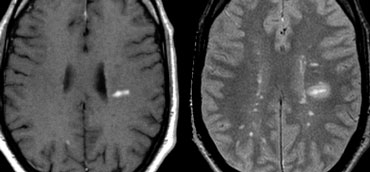
On the far left a new T2 hyperintense lesion on a follow-up examination 3 months after the first clinical event.
The images on the right show a new enhancing lesion on a follow up scan.
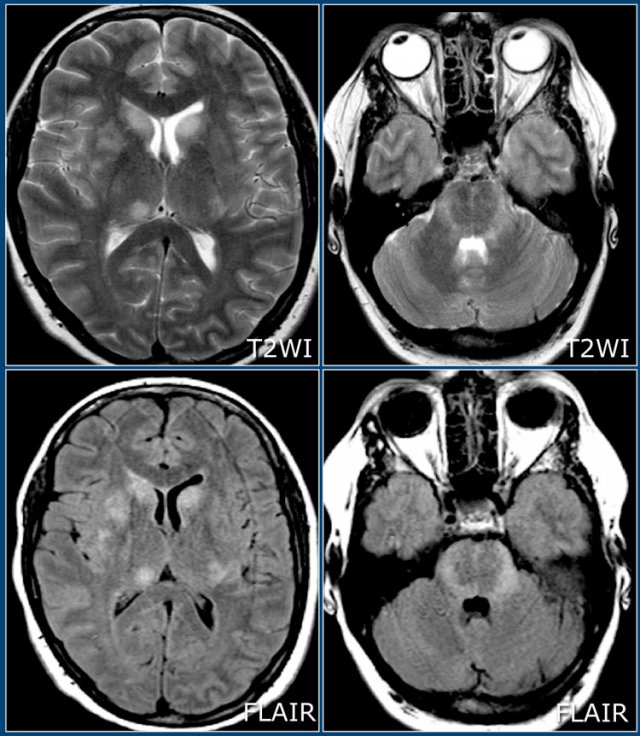
Typical locations of MS lesions
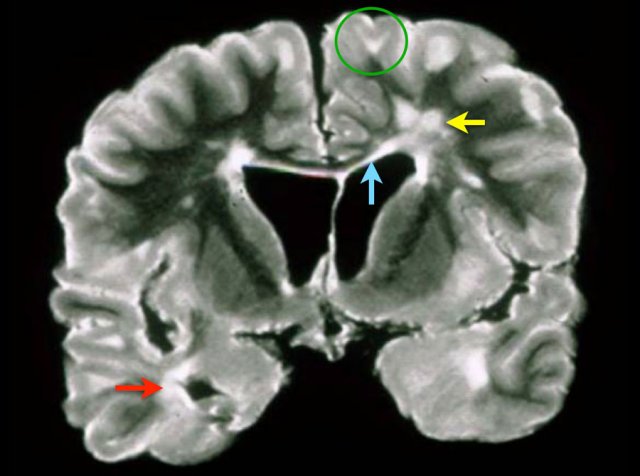
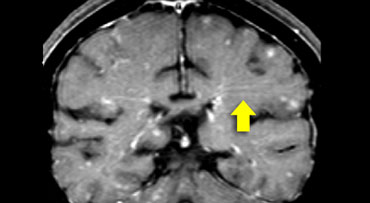
This is a coronal T2 image of a brain specimen with MS involvement. The lesions in the deep white matter (yellow arrow) are nonspecific and can be seen in many diseases.
Typical for MS in this case is:
- Juxtacortical lesions (green circle) - touching the cortex (no intervening WM)
- Periventricular lesions – in direct contact with the ventricles
- Involvement of the corpus callosum (blue arrow)
- Involvement of the temporal lobe (red arrow)
An early diagnosis of MS allows treatment to start earlier and improves prognosis.
This does carry the risk of a false positive diagnosis when MRI findings are misinterpreted.
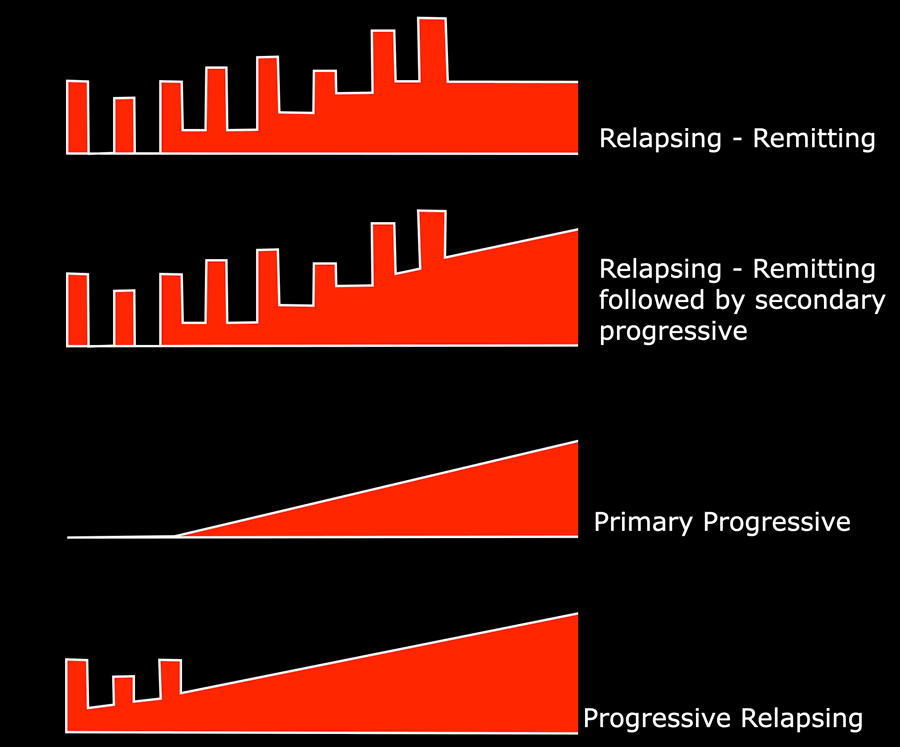
MS phenotypes
Relapsing-remitting (RR)
A multiple sclerosis course characterised by relapses with stable neurological disability between episodes. 85% of cases, F>M.
Secondary progressive (SP)
65% of relapsing-remitting disease becomes secondary progressive.
Primary progressive (PP)
10-15%, M=F
Atypical presentations
Marburg, Schilder, Balo, Devic, tumefactive MS (open-ring sign)
Clinically isolated syndrome (CIS)
A monophasic clinical episode with patient-reported symptoms and objective findings reflecting a focal or multifocal inflammatory demyelinating event in the CNS, developing acutely or subacutely, with a duration of at least 24 h, with or without recovery, and in the absence of fever or infection.
Radiologically isolated syndrome (RIS)
MRI findings strongly suggestive of multiple sclerosis in a patient with no neurological manifestations or other clear-cut explanation.
MR findings in MS
The table summarizes typical MRI-findings in MS.
Even when a patient is clinically suspected of MS, we still must study the WMLs carefully to decide whether these lesions are indeed suggestive of MS, and not incidental age-related findings.
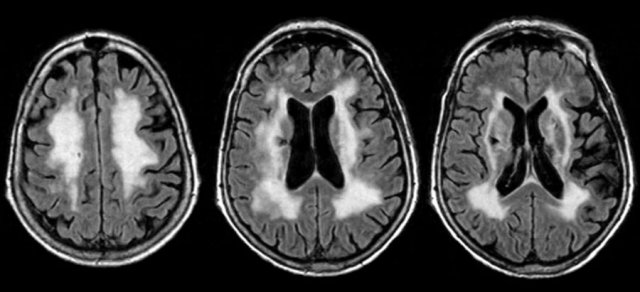
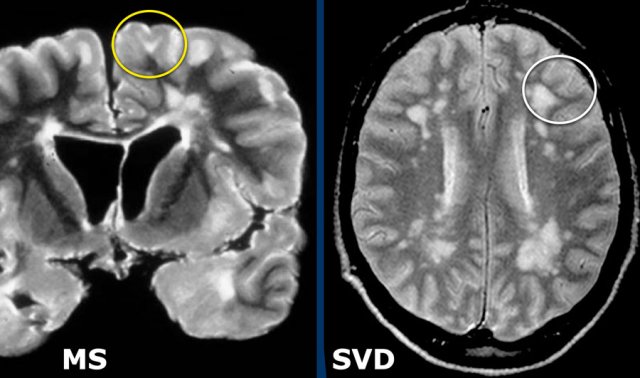 LEFT: involvement of U-fibers in MS. RIGHT: U-fibers are not involved in small vessel disease in a patient with hypertension.
LEFT: involvement of U-fibers in MS. RIGHT: U-fibers are not involved in small vessel disease in a patient with hypertension.
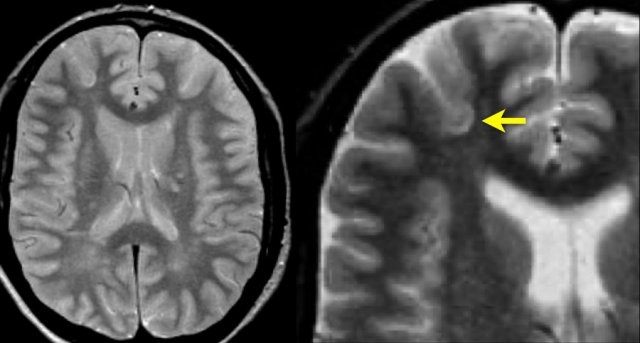
Juxtacortical lesions
Juxtacortical and cortical lesions are specific for MS.
They are adjacent to the cortex and must touch the cortex (yellow circle).
In small vessel disease the U-fibers are typically spared and on T2 and FLAIR there will be a dark band of normal WM between the WML and the bright cortex (white circle).
Do not use the word subcortical to describe this location, because that is a less specific term, indicating a larger area of white matter almost reaching the ventricles.
You really have to look hard to notice juxtacortical lesions because they are difficult to differentiate from the hyperintense cortex.
FLAIR or Double Inversion Recovery (DIR) can help identify them.
The involvement of the U-fibers is best seen on the magnification view.
 LEFT: Typical Dawson finger with enhancement on T1WI. RIGHT: Multiple lesions and edema around enhancing lesion on T2WI.
LEFT: Typical Dawson finger with enhancement on T1WI. RIGHT: Multiple lesions and edema around enhancing lesion on T2WI.
Dawson fingers
Typical findings for MS as seen in this case are:
- Ovoid lesions perpendicular to the ventricles (Dawson fingers).
- Enhancing lesion.
- Multiple lesions adjacent to the ventricles.
Dawson fingers are typical for MS.
They represent areas of demyelination along the small cerebral veins that run perpendicular to the ventricles.
If needed this can be demonstrated using SWI
The enhancement will be present for about one month after the occurrence of a lesion.
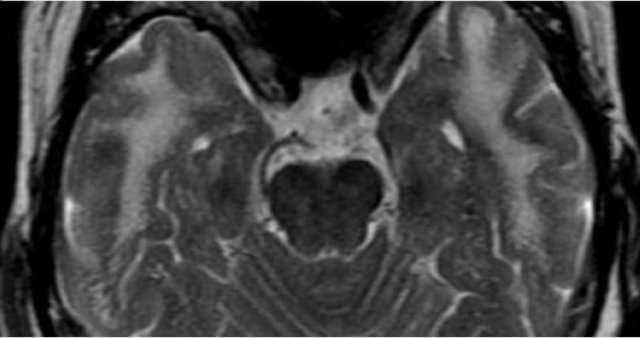
Brainstem lesions
In MS brainstem lesions are typically peripheral.
In small vessel disease there may be involvement of the brainstem, but it is usually symmetrical and central.
Temporal lobe involvement
Temporal lobe involvement is also specific for MS.
In hypertensive encephalopathy, the WMLs mostly found in the frontal and parietal lobes, uncommonly in the occipital lobes and not in the temporal lobes.
Only in CADASIL there is early involvement of the temporal lobes.
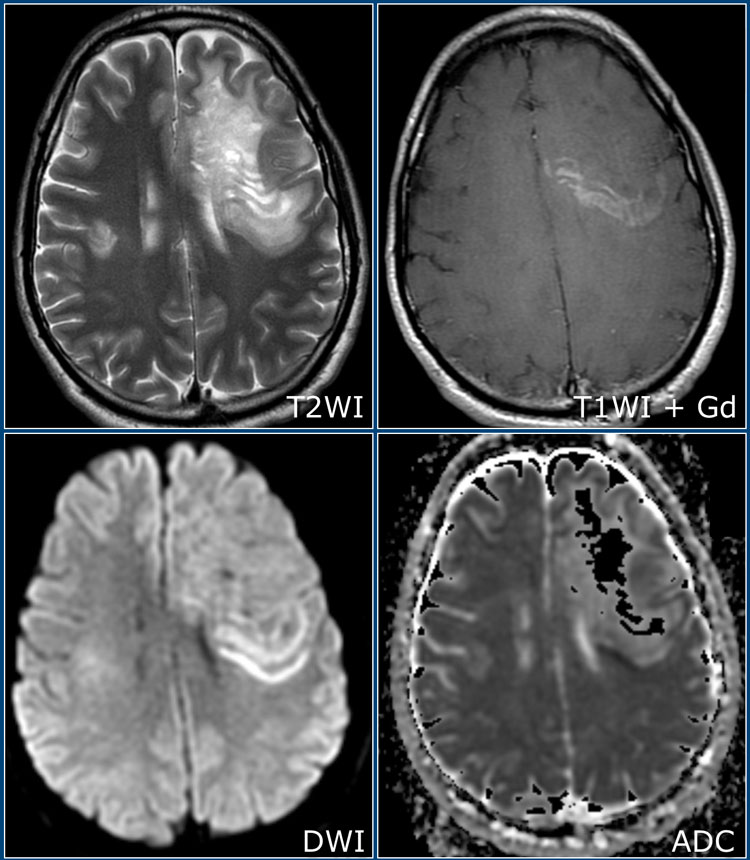
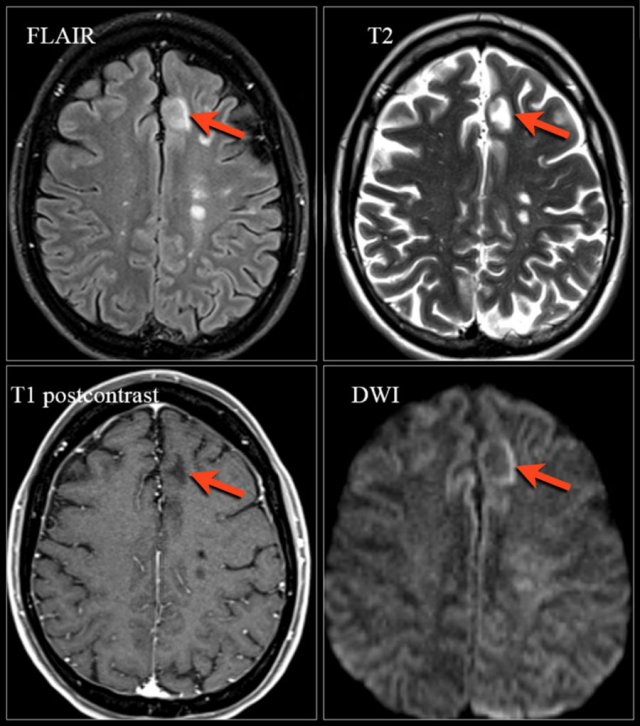
Enhancement
Enhancement is another typical finding in MS.
These are images of a patient who was re-examined 3 months after the first clinical attack.
Typical findings for MS as seen in this case are:
- Multiple enhancing lesions
- Many of these lesions 'touch the cortex' and must be located in the U-fibers.
- These enhancing lesions all are new lesions, since Gadolinium enhancement is only visible for about 1 month.
- So this finding is proof of dissemination in time.
The edema around a new lesion will regress and finally only the center will remain as a hyperintense lesion on T2WI.
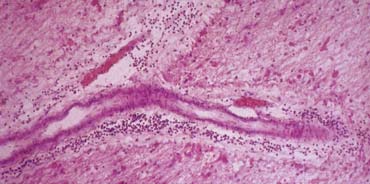
The image shows a specimen with the perivenular inflammation in MS.
MS starts as inflammation around these veins.
In the first four weeks of the inflammation there is enhancement with gadolinium due to local interruption of the blood brain barrier.
First there is homogeneous enhancement but this can change to (open) ring enhancement.
Tumefactive MS
Tumefactive MS is a variant of Multiple Sclerosis.
On MRI it presents as a large intra-parenchymal lesion with usually less mass effect than would be expected for its size.
They may show some peripheral enhancement, often with an incomplete ring unlike gliomas or intraparenchymal abscesses, which typically have a closed-ring enhancement.
These T2W and T1W post-gadolinium images are of a 39-year-old male who presented with subacute onset of hemianopsia.
He was referred for biopsy to differentiate between a glioma or demyelination.
There is an intraparenchymal mass in the right temporal and occital lobe with a hypointense rim on T2, which only partially enhances (open-ring sign) on the postcontrast images.
There is surrounding edema, but relatively little mass effect.
This was a biopsy-proven demyelinating lesion.
The open-ring enhancement pattern with low signal T2 ring and low CBF are all indicative of demyelination.
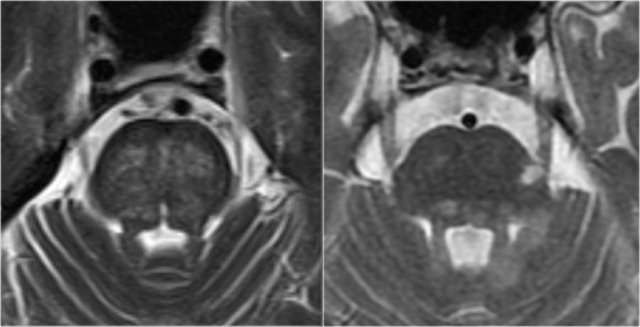
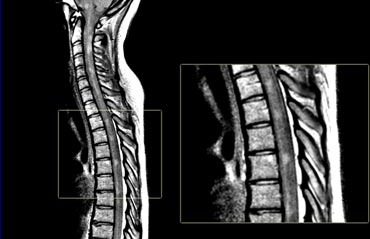
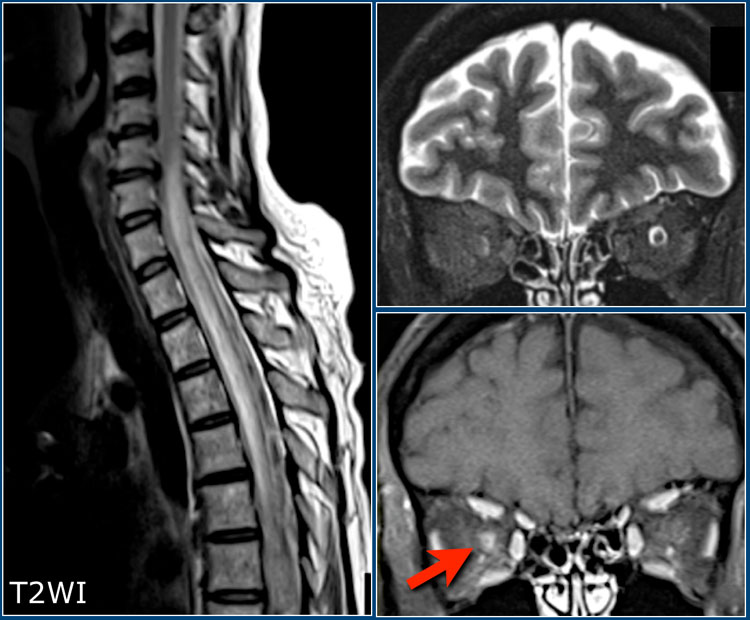
Spinal cord lesions
Typical spinal cord lesions in MS are relatively small and peripherally located.
They are most often found in the cervical cord and are usually less than 2 vertebral segments in length.
A spinal cord lesion together with a lesion in the cerebellum or brainstem is very suggestive of MS.
Spinal cord lesions are uncommon in most other CNS diseases, with the exception of ADEM, sarcoid, and NMOSD.
The images show multiple short-segment lesions in the spinal cord.
On PDW-images the spinal cord has a uniformly low signal intensity (like CSF), which gives the MS lesions a good contrast against the surrounding CSF and normal cord tissue.
Use a 512 matrix and cardiac gating for optimal results.
A good alternative for PDW is STIR.
Make sure to have two different sequences or two different planes.
MS Differential diagnosis
The differential diagnosis for multiple sclerosis (MS) is extensive, encompassing nearly all white matter diseases. While a comprehensive list exists, this discussion focuses on a select few, guided by practical rules:
Rules for Differential Diagnosis
- Clinical Suspicion + Multiple WMLs
If MS is clinically suspected and multiple WMLs are present, the primary differential is MS versus small vessel disease. - Clinical Suspicion + Supportive MRI-findings
If MS is clinically suspected and MRI findings support the diagnosis, uncommon alternatives need not be considered. - No Clinical Suspicion + Incidental WMLs
Do not include MS in the differential diagnosis if:- The clinician does not suspect MS.
- MRI shows incidental white matter lesions (WMLs).
- Rationale: Vascular WMLs are 50–500 times more likely than MS plaques.
Prevalence and a priori chance
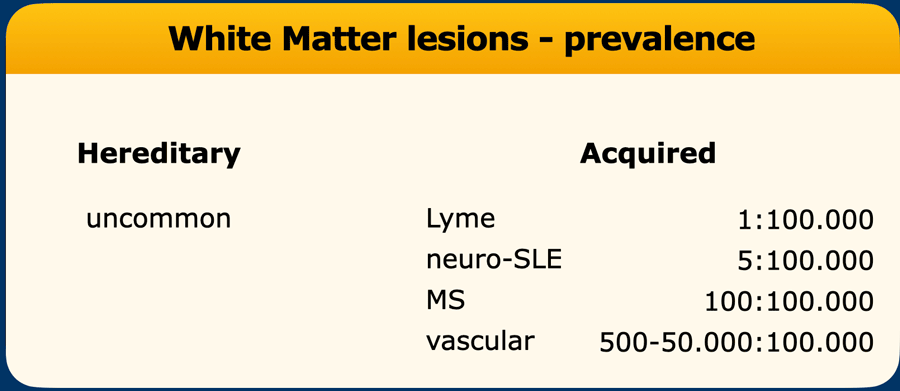
The prevalence of white matter diseases varies dramatically:
- Hereditary Diseases: Individually extremely rare, but collectively less rare—though still far less common than MS.
- CNS Lyme Disease: Extremely rare.
- Incidental WMLs: Most commonly vascular in origin (small vessel disease).
- Up to 50% of patients undergoing MRI for any reason have vascular WMLs.
- Risk factors: Older age, atherosclerosis, hypertension, hypercholesterolemia, diabetes, amyloid angiopathy, hyperhomocysteinemia, atrial fibrillation.
Small vessel disease
The differences between small vessel disease and MS are summarized in the table.
Typical for MS is involvement of corpus callosum, U-fibers, temporal lobes, brainstem, cerebellum and spinal cord.
This pattern of involvement is uncommon in other diseases.
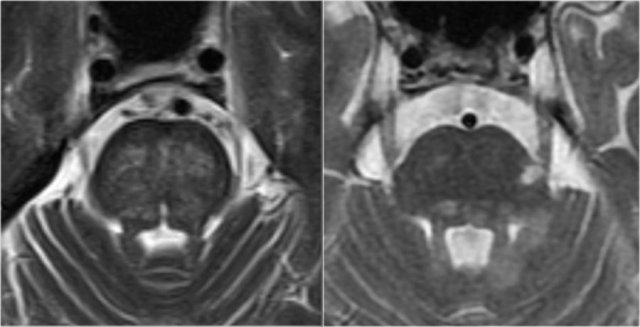
Here we see typical differences in vascular brainstem lesions compared to MS (same images as above).
The image on the left is an axial T2 weighted image illustrating typical vascular brainstem involvement, with a central involvement of the transverse pontine fibers.
The image on the right is an axial T2 weighted image of the brainstem of an MS-patient, showing typical peripherally located white matter lesions, often in or near the trigeminal tract, or bordering the 4th ventricle.
Here is a typical case of small vessel disease.
There is widespread white matter disease with a symmetrical distribution and a lacune in the right basal ganglia.
 Patient with NMOSD showing a longitudinally extensive cord lesion with marked swelling. Bilateral neuritis optica. The diagnosis was confirmed by an AQP4-AB titer of 1:1024.
Patient with NMOSD showing a longitudinally extensive cord lesion with marked swelling. Bilateral neuritis optica. The diagnosis was confirmed by an AQP4-AB titer of 1:1024.
Neuromyelitis Optica
A very important differential to keep in mind, especially in patients with a bilateral optic neuritis and myelitis, is Neuromyelitis Optica Spectrum Disorder (NMOSD), previously called Devic's Disease.
This is a demyelinating disease caused by antibodies against aquaporin or MOG in which the optic nerves and spinal cord are usually involved.
Think of NMOSD when there is longitudinally extensive myelitis (LETM defined as more than 3 vertebral segments) with low T1-signal intensity and swelling of the cord.
On axial images the lesions often involve the central gray matter of the cord.
This is unlike MS, in which the lesions are usually smaller and peripherally located.
Often there are a few T2-lesions in the brain.
Balo's Concentric Sclerosis
Balo's Concentric Sclerosis is an uncommon demyelinating disease.
It is a progressive variant of multiple sclerosis characterized by alternative bands of demyelination and myelin preservation, with a laminated onion-skin configuration.
The T2 and post-contrast T1W images show a large lesion in the left hemisphere with alternating T2-hyperintense and isointense bands.
On the T1W images after gadolinium there is alternating linear enhancement.
Note that the outermost band shows diffusion restriction.
There is a smaller, similar lesion on the right.
ADEM
Acute Disseminated Encephalomyelitis (ADEM) is another important differential diagnosis of MS.
This is a monophasic, immune-mediated demyelinating disease which often presents in children following an infection or vaccination.
Many of the patients have MOG antibodies (MOG= Myelin oligodendrocyte glycoprotein).
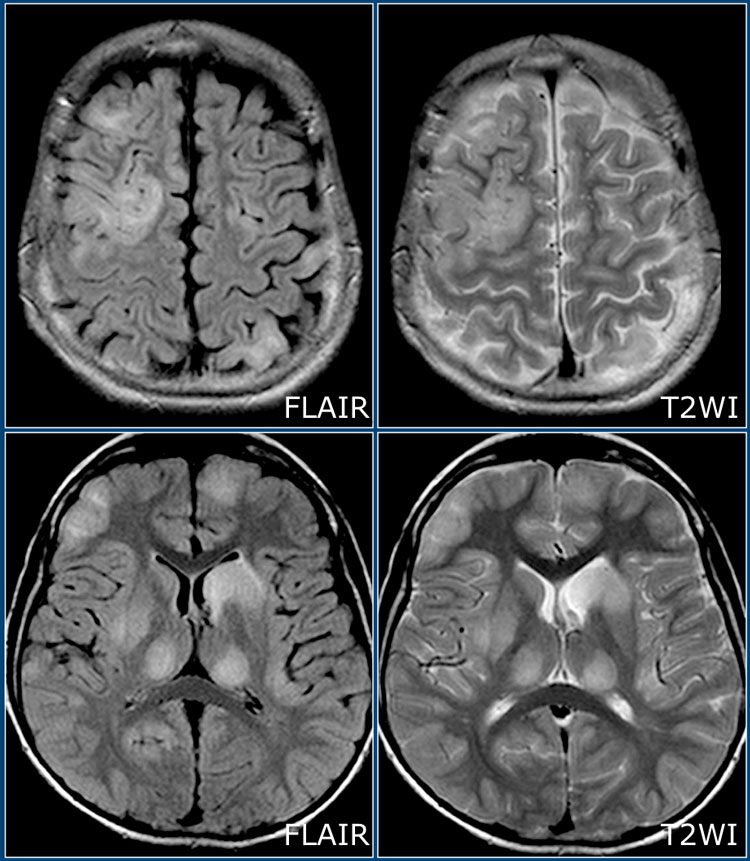
On MRI there are often diffuse and relatively symmetrical lesions in the supra-and infratentorial white matter which may enhance simultaneously.
There often is preferential involvement of the cortical gray matter and the deep gray matter of the basal ganglia and thalami.
Here we have axial FLAIR and T2W-images of a young patient with ADEM - notice the extensive involvement of the cortical and gray matter, including the thalamus.
Here another case of ADEM.
Notice the involvement of the basal ganglia and the middle cerebral peduncle.
Here another case of ADEM.
Notice the similarity to the other two cases.
The differences between ADEM and MS are summarized in the table.
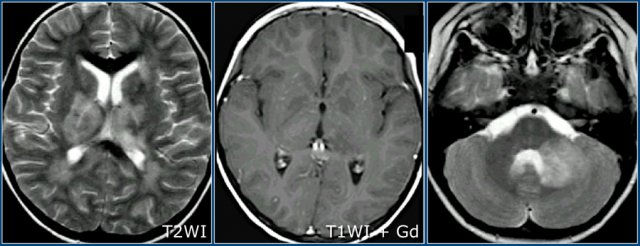
 Natalizumab-associated PML. Images were kindly provided by Bénédicte Quivron CH Jolimont, La Louvière, Belgium
Natalizumab-associated PML. Images were kindly provided by Bénédicte Quivron CH Jolimont, La Louvière, Belgium
Natalizumab-associated PML
Progressive Multifocal Leukoencephalopathy (PML) is a demyelinating disease caused by JC virus in immunosuppressed patients.
Natalizumab is a monoclonal antibody against α4-integrin approved for the treatment of multiple sclerosis due to a positive effect on clinical and magnetic resonance imaging (MRI) outcome measures.
A relatively rare but serious side effect of this drug is a higher risk of developing PML.
The diagnosis of PML according to diagnostic criteria is based on the clinical presentation, the identification of JCV DNA in the CNS (e.g., in the cerebrospinal fluid) and imaging findings preferably on MRI.
Compared to other PML populations such as HIV, the imaging findings in natalizumab-associated PML have been described as heterogeneous and fluctuating.
Key imaging signs are:
- Focal or multifocal lesions in the supratentorial subcortical white matter involving the U-fibers and the cortical grey matter, less frequently posterior fossa and deep grey matter
- T2-hyperintense (occasionally with small focal lesions in the vicinity of the main lesion)
- T1-isointese or hypointense depending on the degree of demyelination
- In approximately 30% of the patients PML lesions can show contrast enhancement.
- High signal intensity on DWI particularly in the border of the lesions reflecting active infection and swelling of the white matter cells.
It can be difficult to differentiate progressive MS from natalizumab-associated PML.
See table for differences in imaging.
How to image natalizumab-associated PML:
- FLAIR has the highest sensitivity in the detection of PML lesions
- T2-weighted sequences can show certain aspects of PML lesions (eg, microcysts)
- T1 with and without contrast is helpful to assess the degree of demyelination and signs of inflammation.
- DWI is useful for the detection of active infection.
Further information: www.MS-PML.org
Sarcoid
Sarcoid has surpassed neurosyphilis as the great mimicker.
The distribution of the lesions in this case is quite similar to MS.
Besides lesions in the deep WM, there are some juxtaventricular lesions and even Dawson finger-like lesions.
The final diagnosis however was sarcoid.
Another typical finding in this case is the linear enhancement (yellow arrow).
This is due to inflammation along the Virchow Robin spaces.
This is also a form of leptomeningeal enhancement.
This explains why sarcoid has a similar distribution to MS: the Virchow Robin spaces follow the small penetrating veins, which are involved in MS.
CADASIL
CADASIL is short for cerebral autosomal dominant arteriopathy with subcortical infarcts and leukencephalopathy.
It is an inherited small vessel disease.
Clinical clues: migraine, dementia and family history.
Key finding: subcortical lacunar infarcts with small cystic lesions and leukoencephalopathy in young adults.
Localizations of the WMLs in the anterior temporal pole and external capsule have a high specificity.
MRI protocol
MS Brain Protocol
Indications for MRI of the brain are (Reference: 2021 MAGNIMS–CMSC–NAIMS consensus recommendations on the use of MRI in patients with multiple sclerosis):
- Clinically isolated syndrome suggestive of MS to show dissemination in time or space in order to fulfill the McDonald criteria
- Patients with MS to determine the prognosis or response to therapy
- To screen for opportunistic infections in patients receiving immunosuppressive treatment (for example development of Progressive Multifocal Leukoencephalopthy in patients using natalizumab).
Gadolinium is administered at the start of the examination because the longer you wait the more enhancement you will see on the T1W images (MS lesions are not spontaneously bright on T1-weighted images without contrast administration).
The sagittal FLAIR is ideal for detection of lesions in the corpus callosum and the 3D sequence allows for better detection of smaller and juxtacortical lesions.
The T2W scan is preferably conventional SE or FSE.
Finally the axial T1W-images are made after about 15 minutes to provide optimal contrast enhancement.
MS Spinal cord Protocol
Indications for MRI of the spinal cord are (Reference: Spinal cord involvement in multiple sclerosis and neuromyelitis optica spectrum disorders):
- Cord symptoms
- To gain specificity in case of non-specific brain lesions
Gadolinium is less relevant when only the spinal cord is examined and is only administered when other diagnoses are considered (e.g. sarcoid) .
The most sensitive sequence is the PD or STIR.
FLAIR should not be used in the spinal cord as it will only demonstrate 10% of the lesions.
MR timing in monitoring MS
The table shows a scheme on how to follow-up a patient with MS based on the 2021 MAGNIMS-CMSC-NAIMS consensus recommendations on the use of MRI in patients with multiple sclerosis.
Consider the following modifications in the protocol:
- Shorter follow-up MRI (i.e, 6 months instead of every year) if substantial isolated MRI activity or isolated clinical activity.
- Longer interval between initial scan and new baseline in patients treated with disease modifying treatments (eg, up to 9 months instead of 3-56 months with glatiramer acetate and until completion of the full initial course with induction therapies).
- Consider gadolinium administration in every patients with highly active disease.
- Less frequent MRI in further follow-ups in clinically stable patients treated with interferon beta or glatiramer acetate instead of every year while on treatment.