Appendicitis and Mimics
Alternative nonsurgical diagnoses at sonography and CT
Adriaan van Breda Vriesman M.D. and Julien Puylaert M.D.
Radiology Department, Rijnland Hospital, Leiderdorp and Medical Centre Haaglanden, the Hague, the Netherlands
Publicationdate
Introduction
In this overview we focus on nonsurgical appendicitis-mimicking diseases.
A correct imaging diagnosis prevents an unnecessary operation or costful in-hospital observation.
For critical comments and additional remarks: j.puylaert@gmail.com
the Appendix
Normal Appendix
Sonography and CT allow direct visualization of the normal or inflamed appendix.
The normal appendix can be identified in 67-100% of patients without appendicitis who undergo CT [1].
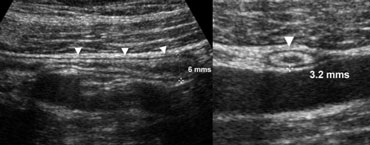 Fig. 1.- 34-year-old healthy volunteer with a normal appendix. A and B, longitudinal (A) and transverse (B) sonogram, showing the appendix (arrowheads) with a diameter less than the 7 mm cut-off point, surrounded by normal noninflamed fat.
Fig. 1.- 34-year-old healthy volunteer with a normal appendix. A and B, longitudinal (A) and transverse (B) sonogram, showing the appendix (arrowheads) with a diameter less than the 7 mm cut-off point, surrounded by normal noninflamed fat.
At sonography the normal appendix is less frequently visualized, with results varying between 0-82% [1], reflecting the operator dependency of sonography.
One of the most important imaging criteria in the evaluation of appendicitis is the outer diameter of the appendix.
Although an overlap of appendiceal diameters in normal and inflamed appendices has been reported, a threshold value of 6-7 mm is most commonly used [1]. (Fig. 1).
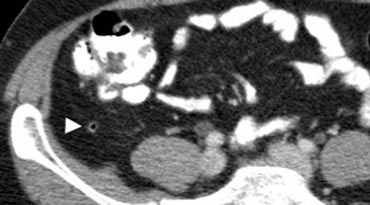 Fig. 2. A 50 year old man with a normal appendix. Unenhanced CT shows an air-filled nondistended appendix (arrowhead) with homogeneous periappendiceal fat without fat-stranding.
Fig. 2. A 50 year old man with a normal appendix. Unenhanced CT shows an air-filled nondistended appendix (arrowhead) with homogeneous periappendiceal fat without fat-stranding.
A normal appendix has a maximum outer diameter of 6 mm, is surrounded by homogeneous non-inflamed fat, and often contains intraluminal gas [2] (Fig. 2).
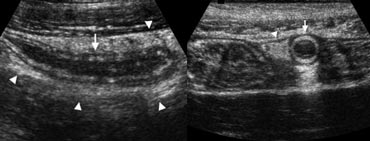 Fig. 3. A 19 year old woman with appendicitis. Longitudinal and transverse sonogram show an enlarged appendix (arrows) surrounded by hyperechoic inflamed fat (arrowheads).
Fig. 3. A 19 year old woman with appendicitis. Longitudinal and transverse sonogram show an enlarged appendix (arrows) surrounded by hyperechoic inflamed fat (arrowheads).
Appendicitis
An inflamed appendix has a diameter larger than 6 mm, and is usually surrounded by hyperechoic inflamed fat at sonography (Fig. 3a).
Other strongly supportive signs of inflammation include the presence of an appendicolith, cecal apical thickening
Another supportive sign for appendicitis is hypervascularity of the appendix wall on color Doppler sonography [1] (Fig. 3b).
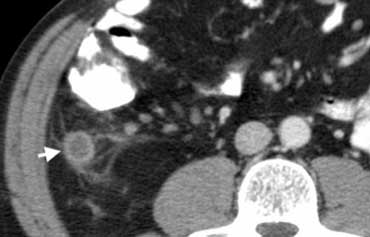 Fig. 4.- 43-year-old man with appendicitis. Contrast-enhanced CT depicts a fluid-filled distended appendix (arrow) with periappendiceal fat-stranding.
Fig. 4.- 43-year-old man with appendicitis. Contrast-enhanced CT depicts a fluid-filled distended appendix (arrow) with periappendiceal fat-stranding.
At CT the inflamed appendix is surrounded by fat-stranding (Fig. 4).
Gastrointestinal nonsurgical mimics of Appendicitis
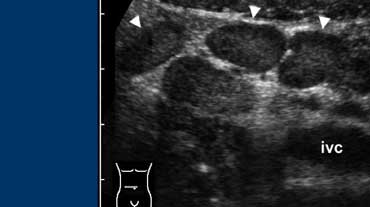 Fig. 5. A 14 year old boy with mesenteric adenitis. Sonogram of the right lower quadrant shows a cluster of enlarged mesenteric lymph nodes (arrowheads). The appendix was normal (not shown) and no other abnormalities were found.
Fig. 5. A 14 year old boy with mesenteric adenitis. Sonogram of the right lower quadrant shows a cluster of enlarged mesenteric lymph nodes (arrowheads). The appendix was normal (not shown) and no other abnormalities were found.
Mesenteric adenitis
Mesenteric adenitis has been reported to be the second most common cause of right lower quadrant pain after appendicitis, accounting for 2-14% of the discharge diagnoses in patients with a clinical suspicion of appendicitis [3].
It is defined as a benign self-limiting inflammation of right-sided mesenteric lymph nodes without an identifiable underlying inflammatory process, occurring more often in children than in adults.
Sonography and CT show clustered adenopathy (Fig. 5).
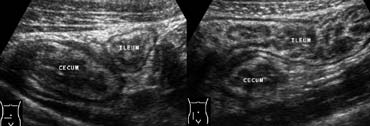 Fig 6.- 39-year-old man with bacterial ileocecitis. A and B, Sonography shows moderate mural thickening of the terminal ileum and cecum, surrounded by normal noninflamed fat. Moderate mesenteric lymphadenopathy was also present (not shown).
Fig 6.- 39-year-old man with bacterial ileocecitis. A and B, Sonography shows moderate mural thickening of the terminal ileum and cecum, surrounded by normal noninflamed fat. Moderate mesenteric lymphadenopathy was also present (not shown).
Bacterial ileocecitis
Because adenopathy also frequently occurs with appendicitis, the normal appendix must be confidently visualized on imaging studies before assigning a diagnosis of mesenteric adenitis. Infectious enterocolitis may cause mild symptoms resembling a common viral gastroenteritis, but it may also clinically present with features indistinguishable from appendicitis [4].
This latter presentation may occur in bacterial ileocecitis, caused by Yersinia, Campylobacter, or Salmonella. Imaging studies show mural thickening of the terminal ileum and cecum without inflammation of the surrounding fat (Fig. 6), and moderate mesenteric adenopathy.
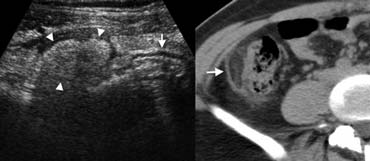 Fig. 7.- 29-year-old woman with epiploic appendagitis. A, Sonography of the right lower quadrant reveals a hyperechoic inflamed fatty mass (arrowheads) adjacent to the colon (arrow), at the spot of maximum tenderness. B, On unenhanced CT the fatty lesion contains a characteristic hyperattenuating ring (arrow) corresponding to thickened visceral peritoneal lining.
Fig. 7.- 29-year-old woman with epiploic appendagitis. A, Sonography of the right lower quadrant reveals a hyperechoic inflamed fatty mass (arrowheads) adjacent to the colon (arrow), at the spot of maximum tenderness. B, On unenhanced CT the fatty lesion contains a characteristic hyperattenuating ring (arrow) corresponding to thickened visceral peritoneal lining.
Epiploic appendagitis
Epiploic appendages are small adipose protrusions from the serosal surface of the colon. An epiploic appendage may undergo torsion and secondary inflammation, causing focal abdominal pain that simulates appendicitis when located in the right lower quadrant.
Epiploic appendagitis is a self-limiting disease that has been reported in approximately 1% of patients clinically suspected of having appendicitis [5].
Sonography and CT depict an inflamed fatty mass adjacent to the colon (Fig. 7), containing a characteristic hyperattenuating ring of thickenend visceral peritoneal lining on CT [5].
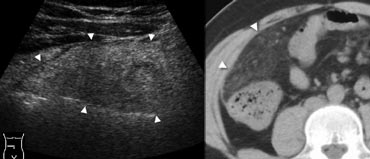 Fig. 8.- 41-year-old man with omental infarction. A, Sonography of the right middle abdomen shows a large area of inflamed intraperitoneal fat (arrowheads). B, Unenhanced CT depicts the lesion as a cake-like area of dense inflamed omental fat (arrowheads), larger than in epiploic appendagitis and lacking a hyperattenuating ring.
Fig. 8.- 41-year-old man with omental infarction. A, Sonography of the right middle abdomen shows a large area of inflamed intraperitoneal fat (arrowheads). B, Unenhanced CT depicts the lesion as a cake-like area of dense inflamed omental fat (arrowheads), larger than in epiploic appendagitis and lacking a hyperattenuating ring.
Omental infarction
Omental infarction has a pathophysiology and clinical presentation similar to that of epiploic appendagitis, with the infarcted fatty tissue being a right-sided segment of the omentum. Imaging shows a cakelike inflamed fatty mass (Fig. 8), larger than in epiploic appendagitis and lacking a hyperattenuating ring on CT.
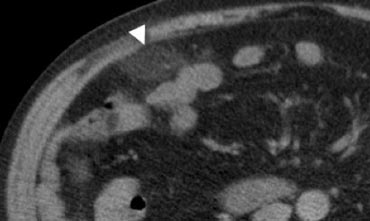 Fig. 9.- 47-year-old woman with acute right lower quadrant pain. Unenhanced CT shows an ovoid inflamed fatty mass (arrowhead) with normal regional bowel loops. The shape and size of the lesion suggests epiploic appendagitis, but the lesion does not contain a hyperattenuating ring. In this case, it is difficult to discriminate between epiploic appendagitis or a small omental infarction.
Fig. 9.- 47-year-old woman with acute right lower quadrant pain. Unenhanced CT shows an ovoid inflamed fatty mass (arrowhead) with normal regional bowel loops. The shape and size of the lesion suggests epiploic appendagitis, but the lesion does not contain a hyperattenuating ring. In this case, it is difficult to discriminate between epiploic appendagitis or a small omental infarction.
In some cases it may be difficult to distinguish epiploic appendagitis from omental infarction (Fig. 9), however, this distinction has no clinical importance as both have a similar benign natural history [5].
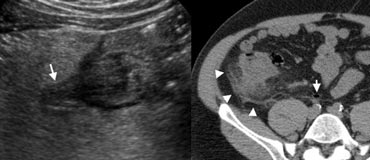 Fig. 10.- 51-year-old man with right-sided colonic diverticulitis. A, Unenhanced CT shows extensive with fat-standing along the cecal wall (arrowheads), and a normal appendix (arrow). B, Sonography reveals the cause of the inflammation by depicting an inflamed cecal diverticulum (arrow) centred in the hyperechoic fat.
Fig. 10.- 51-year-old man with right-sided colonic diverticulitis. A, Unenhanced CT shows extensive with fat-standing along the cecal wall (arrowheads), and a normal appendix (arrow). B, Sonography reveals the cause of the inflammation by depicting an inflamed cecal diverticulum (arrow) centred in the hyperechoic fat.
Right-sided colonic diverticulitis
Right-sided colonic diverticulitis may clinically mimic appendicitis or cholecystitis, though the patient's history is generally more protracted. In contrast to sigmoid diverticula, right-sided colonic diverticula are usually true diverticula, that is, outpouchings of the colonic wall containing all layers of the wall.
This may possibly explain the essentially benign self- limiting character of right-sided diverticulitis [6].
Sonography and CT findings consist of inflammatory changes in the pericolic fat with segmental thickening of the colonic wall, at the level of an inflamed diverticulum (Fig. 10).
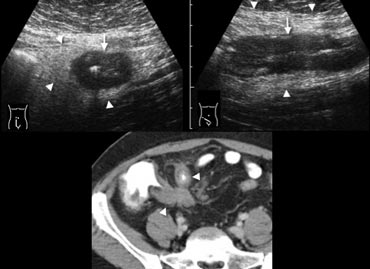 Fig. 11.- 28-year-old man with acute ileocecal Crohn disease.A and B, Sonography shows transmural wall thickening of the terminal ileum (arrows) in longitudinal (A) and transverse (B) section, with hyperechoic inflammatory changes of the surrounding fat (arrowheads).C, Contrast-enhanced CT confirms the wall thickening and luminal narrowing of the terminal and pre-terminal ileum (arrowheads), with regional fat-stranding.
Fig. 11.- 28-year-old man with acute ileocecal Crohn disease.A and B, Sonography shows transmural wall thickening of the terminal ileum (arrows) in longitudinal (A) and transverse (B) section, with hyperechoic inflammatory changes of the surrounding fat (arrowheads).C, Contrast-enhanced CT confirms the wall thickening and luminal narrowing of the terminal and pre-terminal ileum (arrowheads), with regional fat-stranding.
Crohn disease
Crohn disease often causes long-standing symptoms, but up to one third of patients with ileocecal Crohn disease present with initial symptoms so acute that they are misdiagnosed as appendicitis [7].
In the acute active phase of ileocecal Crohn disease,
imaging shows transmural bowel wall thickening, often predominantly of the submucosal
layer, with frequent inflammatory changes of the surrounding fat (Fig. 11).
Uncomplicated
Crohn disease can initially be treated with anti-inflammatory drugs.
Other nonsurgical mimics of appendicitis
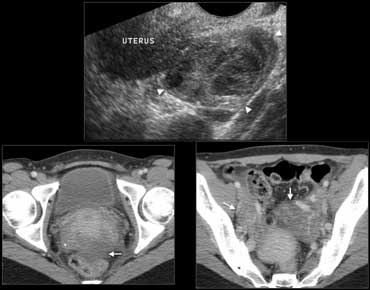 Fig. 12.- 39-year-old woman with pelvic inflammatory disease.A, Endovaginal sonography shows an inhomogeneously enlarged right ovary (arrowheads).B and C, Contrast-enhanced CT shows enlargement of the ovaries (B, arrows) with ill-defined contours of the ovaries and uterus, and some free pelvic fluid (C, arrow).
Fig. 12.- 39-year-old woman with pelvic inflammatory disease.A, Endovaginal sonography shows an inhomogeneously enlarged right ovary (arrowheads).B and C, Contrast-enhanced CT shows enlargement of the ovaries (B, arrows) with ill-defined contours of the ovaries and uterus, and some free pelvic fluid (C, arrow).
Gynecologic conditions
Gynecologic conditions such as pelvic inflammatory disease or a hemorrhagic
functional ovarian cyst can cause acute pelvic pain that may simulate appendicitis.
In pelvic
inflammatory disease the imaging findings vary according to the severity of the disease, and
may be normal in early conditions.
In more advanced stages, findings may include
enlargement of the internal genital organs with indistinct contours, and free pelvic fluid (Fig.
12).
In absence of a drainable tubo-ovarian abscess, treatment is medically with antibiotics.
An hemorrhagic ovarian cysts appears as a complicated cyst at sonography and a high-
attenuation adnexal mass at unenhanced CT, and does not require any treatment.
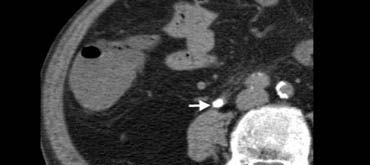 Fig. 13. 77-year-old man with a right ureteral stone.Unenhanced CT shows an obstructing calcification (arrow) within the distal ureteral lumen.
Fig. 13. 77-year-old man with a right ureteral stone.Unenhanced CT shows an obstructing calcification (arrow) within the distal ureteral lumen.
Urolithiasis
Urolithiasis may present with right lower quadrant pain when obstruction is caused by a distal ureteral stone. Unenhanced CT (Fig. 13) is more accurate in detecting ureteral stones than sonography,
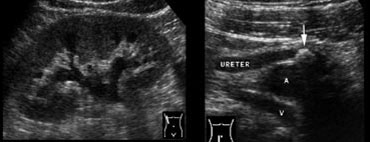 Fig. 14. 40-year-old woman with a ureteral stone.A and B, Sonography shows right-sided hydronephrosis (A), and an obstructing calculus (B, arrow) in the distal ureter at the level of the iliac vessels.
Fig. 14. 40-year-old woman with a ureteral stone.A and B, Sonography shows right-sided hydronephrosis (A), and an obstructing calculus (B, arrow) in the distal ureter at the level of the iliac vessels.
Ultrasound may show both hydronephrosis and hydroureter as signs of obstruction (Fig. 14).
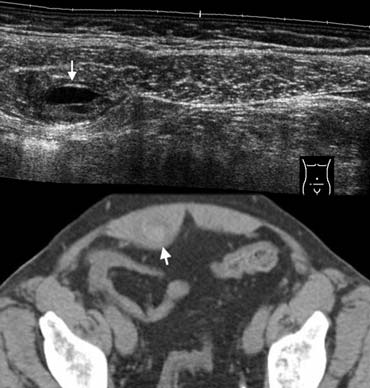 Fig. 15.- 68-year-old woman with a rectus sheath hematoma.A, Sonography depicts a small painfull lesion (arrow) within the sheath of the rectus abdominis muscle in the right lower quadrant. The lesion contains a fluid-fluid level.B, Unenhanced CT depicts the lesion as a partly hyperdense mass (arrow) within the rectus sheath.
Fig. 15.- 68-year-old woman with a rectus sheath hematoma.A, Sonography depicts a small painfull lesion (arrow) within the sheath of the rectus abdominis muscle in the right lower quadrant. The lesion contains a fluid-fluid level.B, Unenhanced CT depicts the lesion as a partly hyperdense mass (arrow) within the rectus sheath.
Rectus sheath hematoma
A rectus sheath hematoma may be easy to diagnose in patients presenting with a
painful palpable mass under anticoagulant therapy, however, small nonpalpable hematomas
may clinically masquerade as appendicitis and also occur in patients without anticoagulantia
[8].
Sonography and CT show a hemorrhagic mass within the sheath of the rectus abdominis
muscle (Fig. 15).
No treatment is required other than adjusting any anticoagulant therapy.
Conclusion
A broad spectrum of nonsurgical diseases may clinically present as appendicitis in
patients without appendicitis.
The radiologist should be aware of the sonographic and CT
features of these alternative disorders, as a correct imaging diagnosis prevents an unwarranted
operation and unnecessary hospital resource use.