Multiple Sclerosis 3.0
2024 McDonald criteria - Diagnosis and differential diagnosis
Frederik Barkhof and Robin Smithuis
Amsterdam University Medical Center and University College London and Alrijne Hospital in Leiden, the Netherlands
Publicationdate
The 2024 McDonald criteria prioritize typical lesions with dissemination in space (DIS) as the diagnostic cornerstone, moving away from the previous requirement for dissemination in time (DIT) to distinguish MS from monophasic inflammatory syndromes.
This shift reflects the impact of modern disease-modifying therapies on diagnostic and treatment paradigms.
Major Revisions:
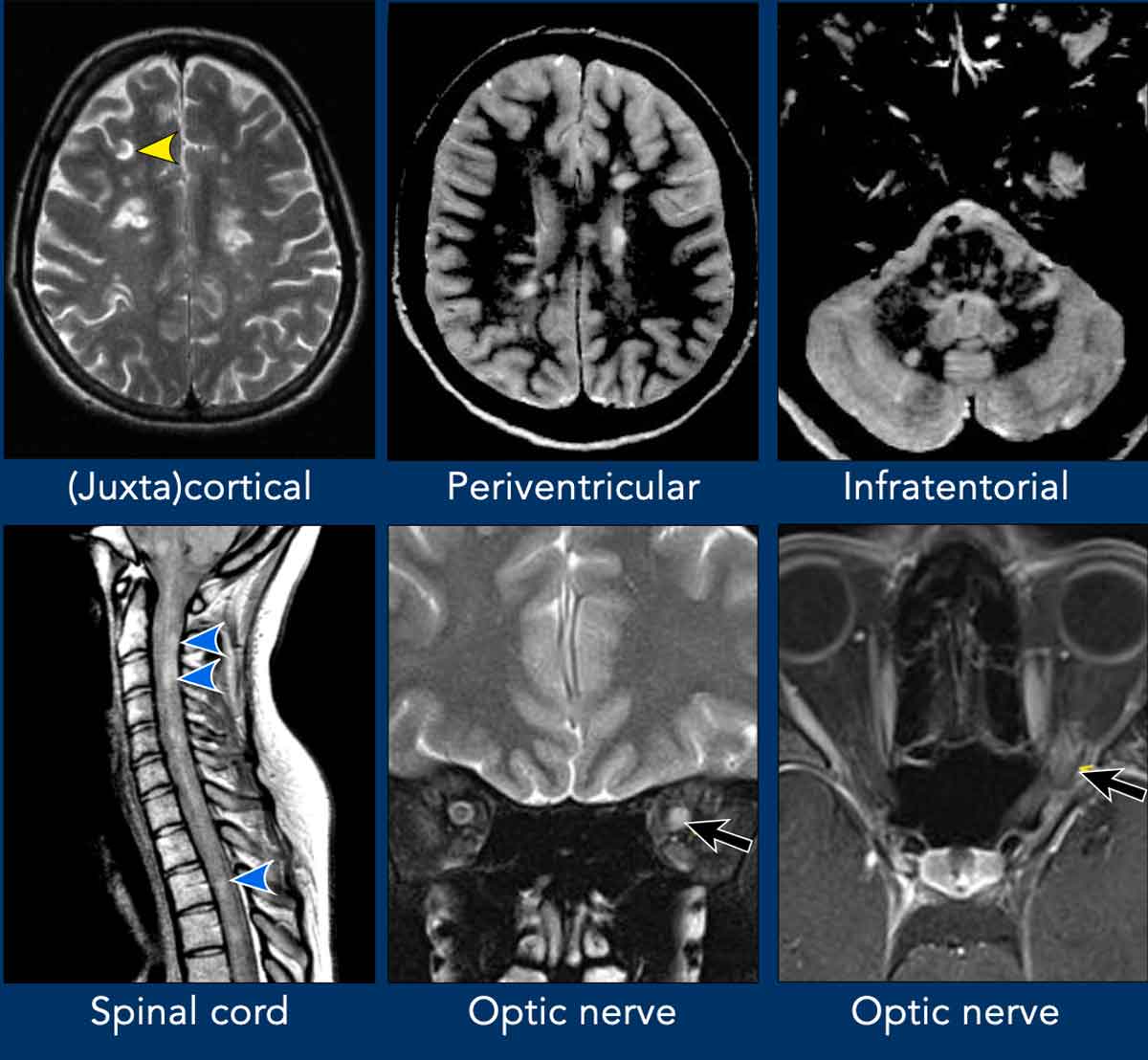
- Optic nerve added as a fifth typical location (alongside periventricular, juxtacortical-cortical, infratentorial, and spinal cord regions).
- DIS criteria met with lesions in ≥ 2 of 5 locations.
- MS diagnosis possible with lesions in ≥ 4 of 5 locations, without further evidence in relapsing or progressive-onset cases.
DIT in many cases no longer mandatory; alternatives include:
- Extensive DIS (≥4/5 regions)
- Central vein sign (CVS) or paramagnetic rim lesions (PRL)
- CSF kappa free-light chain index or oligoclonal bands (OCB)
We will further discuss differentiating MS lesions from other white matter diseases and the role of a priori probability in the differential diagnosis of white matter lesions.
2024 McDonald criteria
Dissemination in space
The role of Dissemination in Space (DIS) has expanded, while Dissemination in Time (DIT) is now less emphasized, enabling earlier MS diagnosis.
DIS criteria are now met when typical lesions are present in ≥2 of 5 anatomical regions, regardless of symptomatology:
- Periventricular
- Cortical/juxtacortical
- Infratentorial brain
- Spinal cord
- Optic nerve
Inclusion of the optic nerve as a fifth typical site enhances diagnostic sensitivity without compromising specificity.
For relapsing and progressive-onset MS (now considered a continuum), diagnosis can be confirmed if ≥4 of 5 anatomical locations show typical lesions, without requiring additional DIT or any other evidence.
In primary progressive MS there is the alternative DIS option of ≥2 spinal cord lesions, allowing diagnosis even in the absence of brain lesions.
Dissemination in time
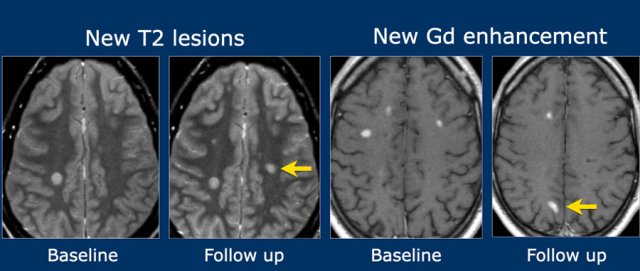
Dissemination in Time (DIT) is demonstrated by either:
- New T2 or gadolinium-enhancing lesion on follow-up MRI, compared to a baseline scan (regardless of baseline timing).
- Simultaneous presence of both gadolinium-enhancing and non-enhancing lesions at any time.
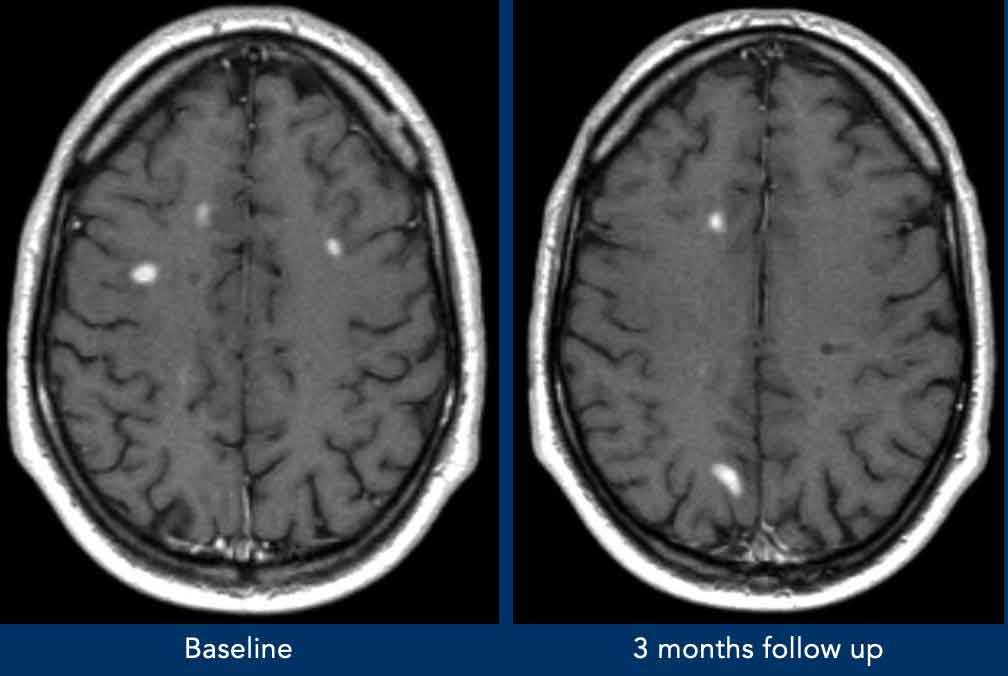
Example:
- Left image: New T2 hyperintense lesion on follow-up MRI, 3 months post-initial clinical event.
- Right images: New enhancing lesion on follow-up scan.
Relapse / Progressive onset
Almost all patient with MS present with relapse remitting or progressive onset MS (typical attack or neurological progression in 12 months or more). Relapse and progressive-onset MS are now regarded to constitute a continuum.
In the 2024 McDonald diagnostic criteria, typical lesions with dissemination in space have become the cornerstone of the diagnosis.
The 2024 criteria mark a dramatic step away from mandatory demonstration of dissemination in time, which was previously needed to distinguish MS from monophasic inflammatory syndromes.
In the setting of relapsing- and progressive-onset MS, the diagnosis of MS can now be established without DIT if at least four of five anatomical locations are affected by typical lesions If less than 4 regions are affected, MS can still be diagnosed, when there is CSF positivity or Central Vein Sign (CVS) or Paramagnetic Rim Lesions (PRL).
There is just one remaining difference for primary progressive MS, where evidence of two or more spinal cord lesions is sufficient to fulfill the criteria for DIS, recognizing the ability to diagnose MS without brain lesions.
Radiologically isolated syndrome
Radiologically isolated syndrome (RIS) is defined by incidental MRI detection of T2-hyperintense lesions in the CNS consistent with multiple sclerosis, but without clinical symptoms.
About half of individuals with RIS develop clinical symptoms within 10 years. RIS in the McDonald 2024 criteria can now be called MS if sufficient evidence is available, without any symptoms, and be treated as MS.
MR findings in MS
Even with clinical suspicion of MS, careful evaluation of white matter lesions is essential to distinguish MS-typical lesions from incidental age-related changes and other diseases like Myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) and Aquaporin-4 antibody-positive Neuromyelitis Optica Spectrum Disorder (AQP4+NMOSD).
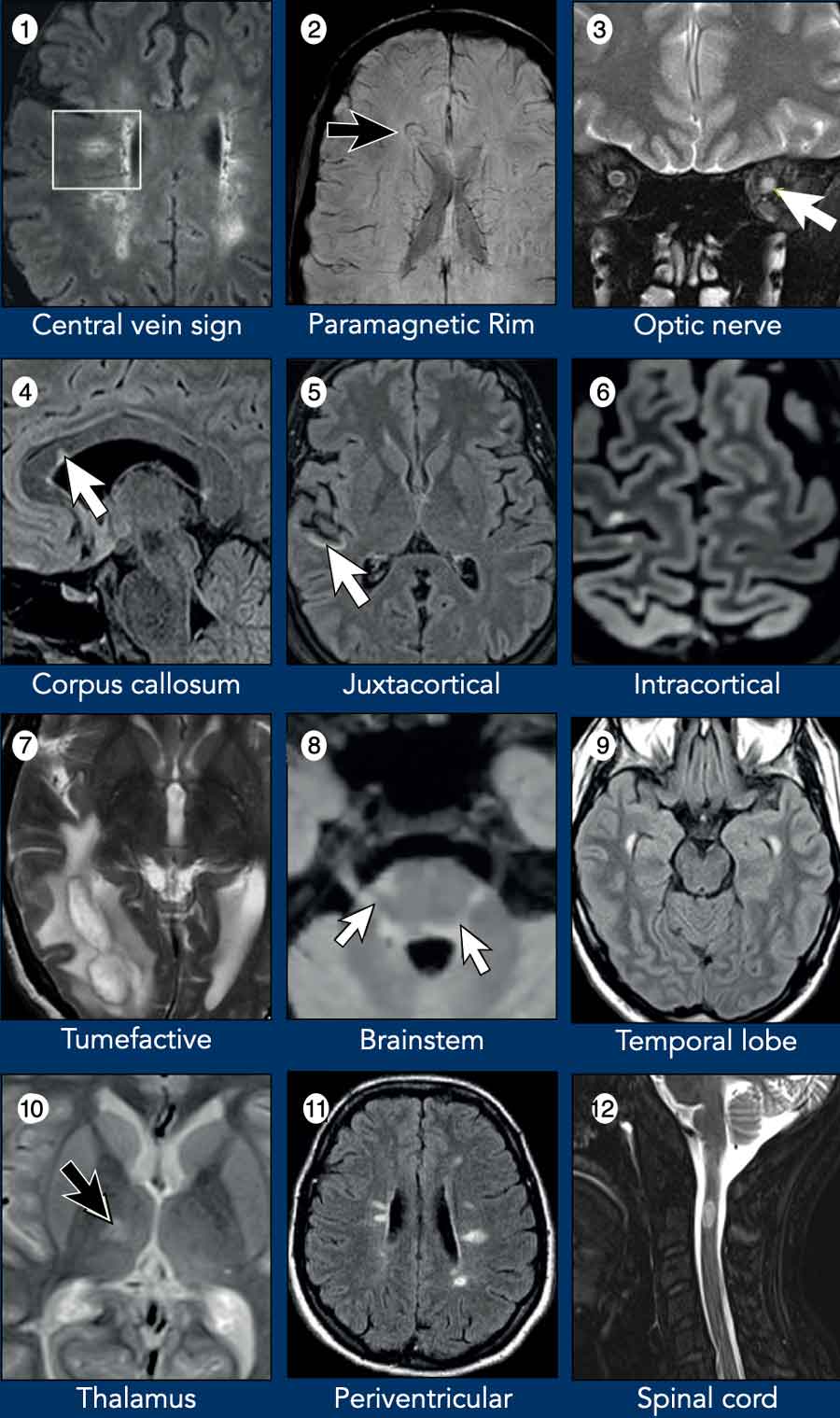
Typical MRI-findings in MS:
- Central vein sign (CVS). The CVS identifies the perivenous distribution of multiple sclerosis lesions. Best visualized with susceptibility-sensitive MRI; new McDonald criterion.
- Paramagnetic rim lesion (PRL). Highly MS-specific; new McDonald criterion.
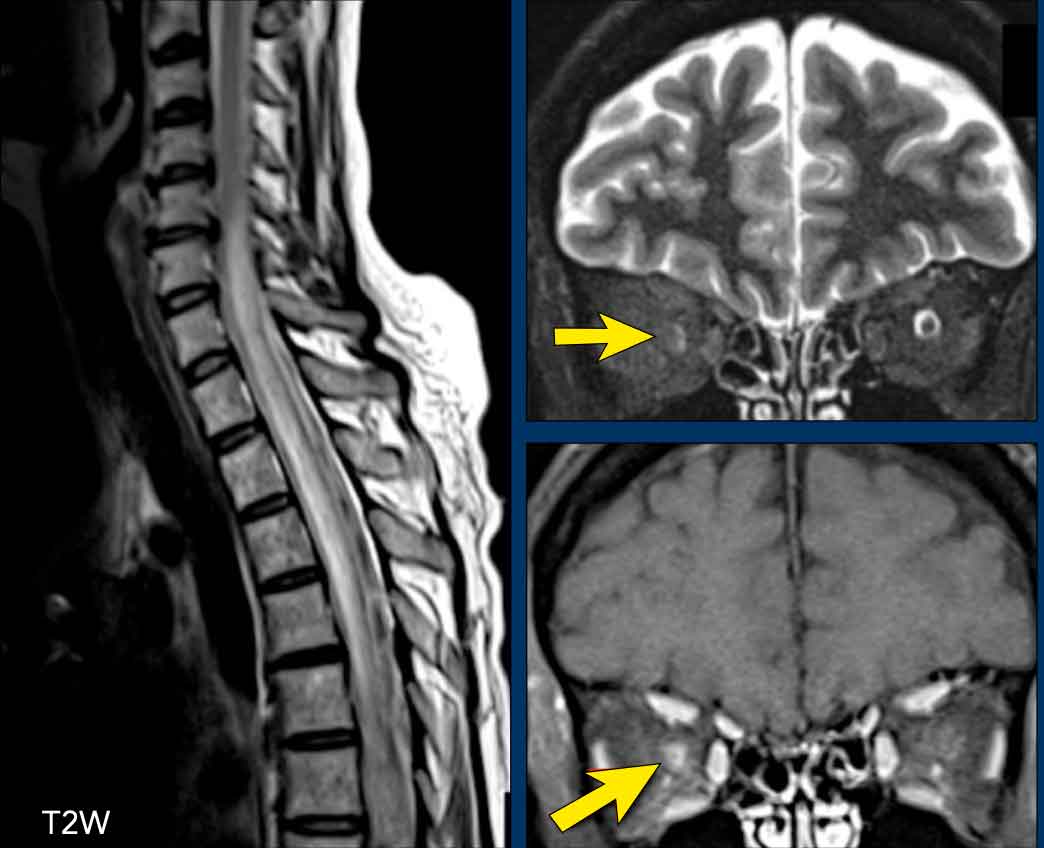
- Optic nerve. Optic neuritis can be used as one of the five anatomical locations for DIS, even in individuals without a clinical history of optic neuritis.
- Corpus callosum. Periventricular lesions include corpus callosum lesions that abut the ventricles.
- Juxtacortical lesions with typical morphology are a hallmark feature of MS. Rarely encountered in other conditions.
- Cortical lesion. Cortical lesions are a hallmark of MS progression and cognitive impairment, best detected with advanced MRI techniques like DIR, 3D FLAIR. Their presence helps differentiate MS from other demyelinating diseases.
- Tumefactive MS with surrounding edema and a dark T2 line around the center. Rare, mimics glioma/metastasis.
- Brainstem. Common; involves midbrain (periaqueductal gray, cerebral peduncles) and pons (surface, border of 4th ventricle and trigeminal tract).
- Anterior temporal lobe lesion. Uncommon in other diseases; also seen in CADASIL.
- Thalamus. MS can involve the deep grey matter (mainly the thalamus).
- Periventricular lesions. Direct contact with lateral ventricles, no intervening normal-appearing white matter.
- Spinal cord. Multiple short-segment lesions typical.
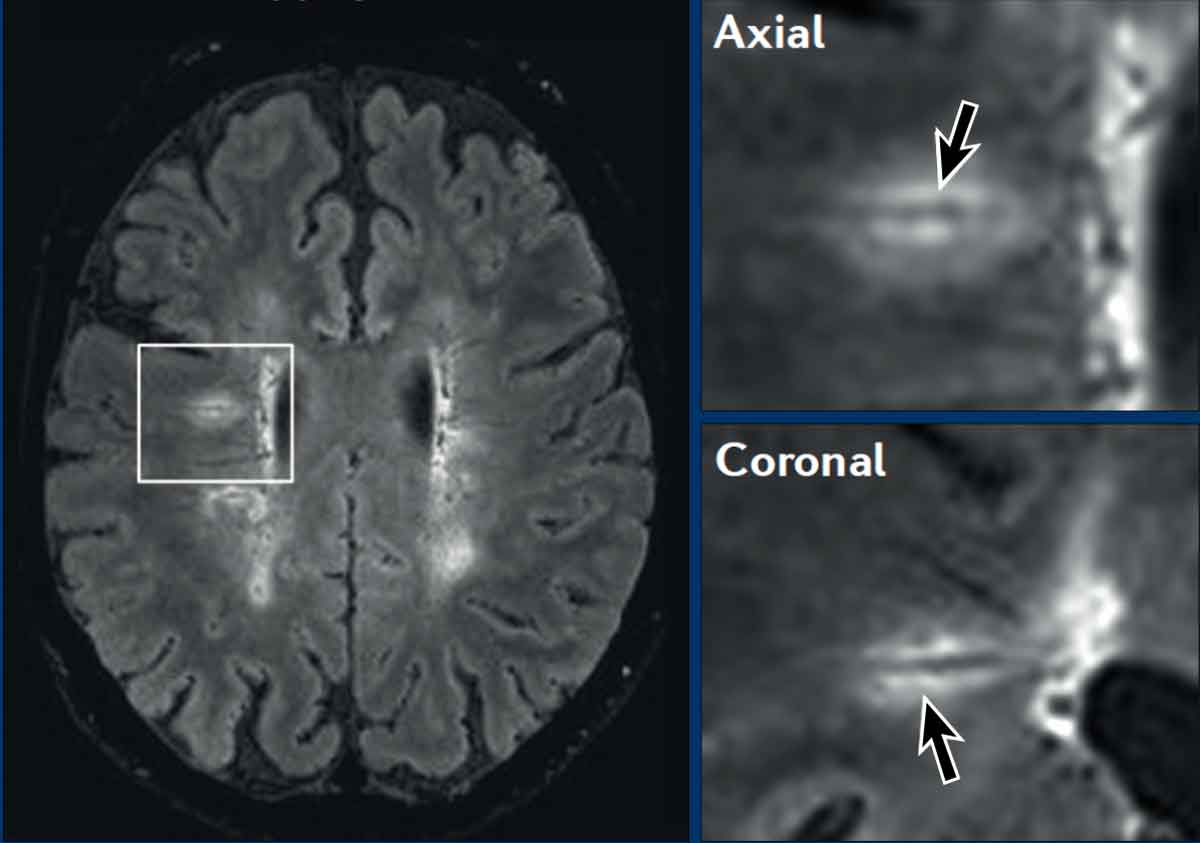
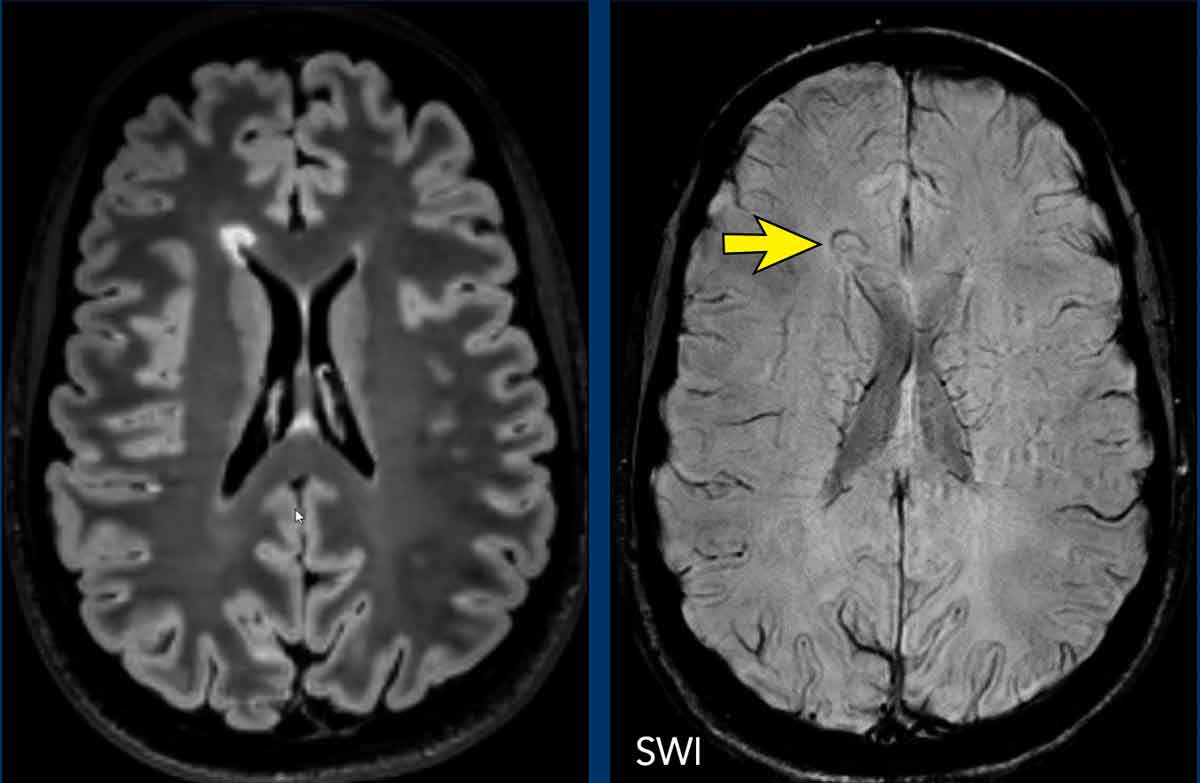
Central vein sign
The central vein sign (CVS) confirms perivenular inflammatory demyelination, a hallmark of MS lesions and can be detected with susceptibility-sensitive MRI sequences..
CVS enhances diagnostic specificity by distinguishing MS lesions from common mimickers of multiple sclerosis, including cerebral small vessel disease, AQP4 + NMOSD, and migraine, that rarely show the presence of a central vein.
In patients fulfilling 2-3 DIS criteria, demonstration of CVS is sufficient for MS diagnosis, obviating the need for additional evidence such as dissemination in time.
Imaging Characteristics
- Appears as a thin (<2 mm) hypointense line or dot
- Best visualized on magnitude susceptibility-weighted images (CVS/PRL protocol)
- Most prevalent in periventricular and deep white matter lesions
Diagnostic Criteria
- Select 6: ≥6 lesions with a central vein
- In patients with ≤10 white matter lesions, CVS in the majority is sufficient for diagnosis.
Paramagnetic rim lesions - PRL
PRL are a distinctive feature of MS, reflecting iron-containing microglia surrounding chronic-active white matter lesions, and portend poor prognosis.
They are highly specific to MS.
Detection of PRL is most efficient using high-field MRI scanners, particularly at 3 T and combining magnitude and phase images (see CVS and PRL protocol).
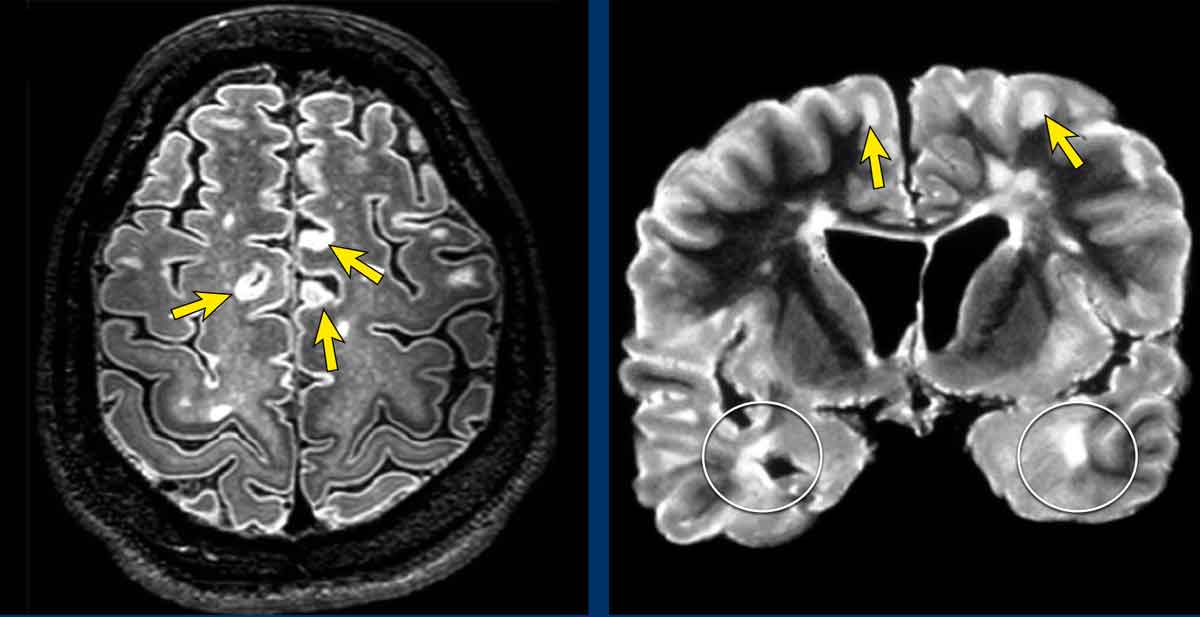
Cortical and Juxtacortical lesions
Juxtacortical and cortical lesions are specific for MS.
They are adjacent to the cortex and must touch the cortex (arrows).
Do not use the word subcortical to describe this location, because that is a less specific term, indicating a larger area of white matter almost reaching the ventricles.
Temporal lobe involvement is also specific for MS (white circles).
In hypertensive encephalopathy, the WMLs mostly found in the frontal and parietal lobes, uncommonly in the occipital lobes and not in the temporal lobes.
Only in CADASIL there is early involvement of the temporal lobes.
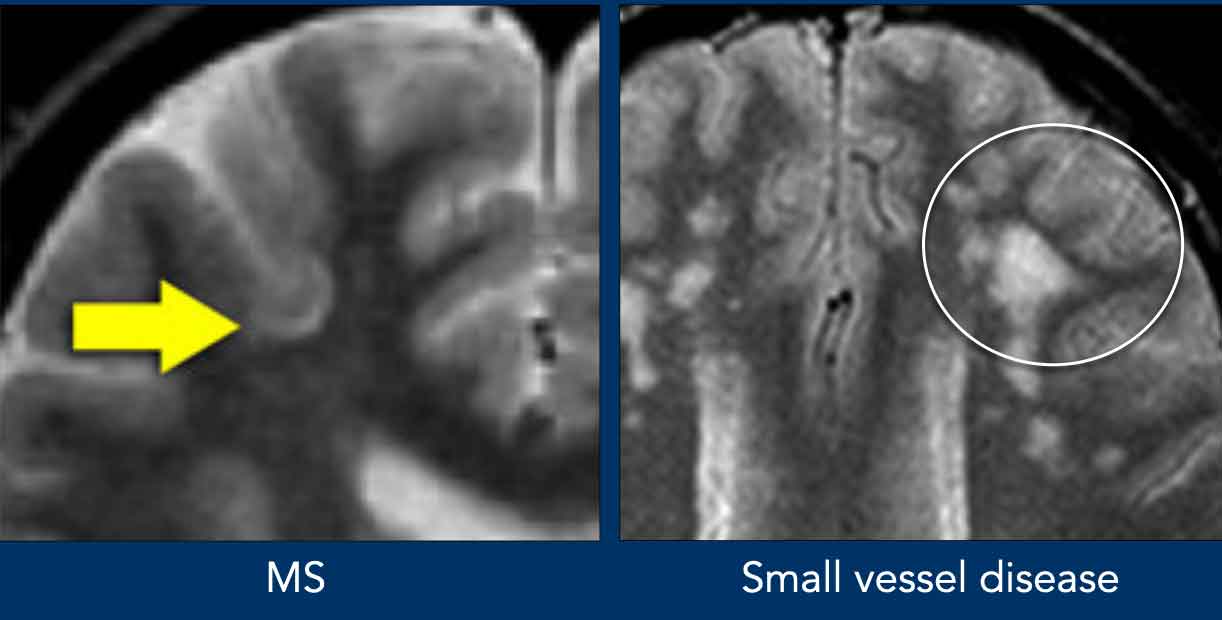
U-fibers
Juxtacortical MS lesion located in the cortex or in the U-fibers (arrow), while the U-fibers are typically spared in small vessel disease (circle).
You really have to look hard to notice juxtacortical lesions because they are difficult to differentiate from the hyperintense cortex.
FLAIR or Double Inversion Recovery (DIR) can help identify these lesions.
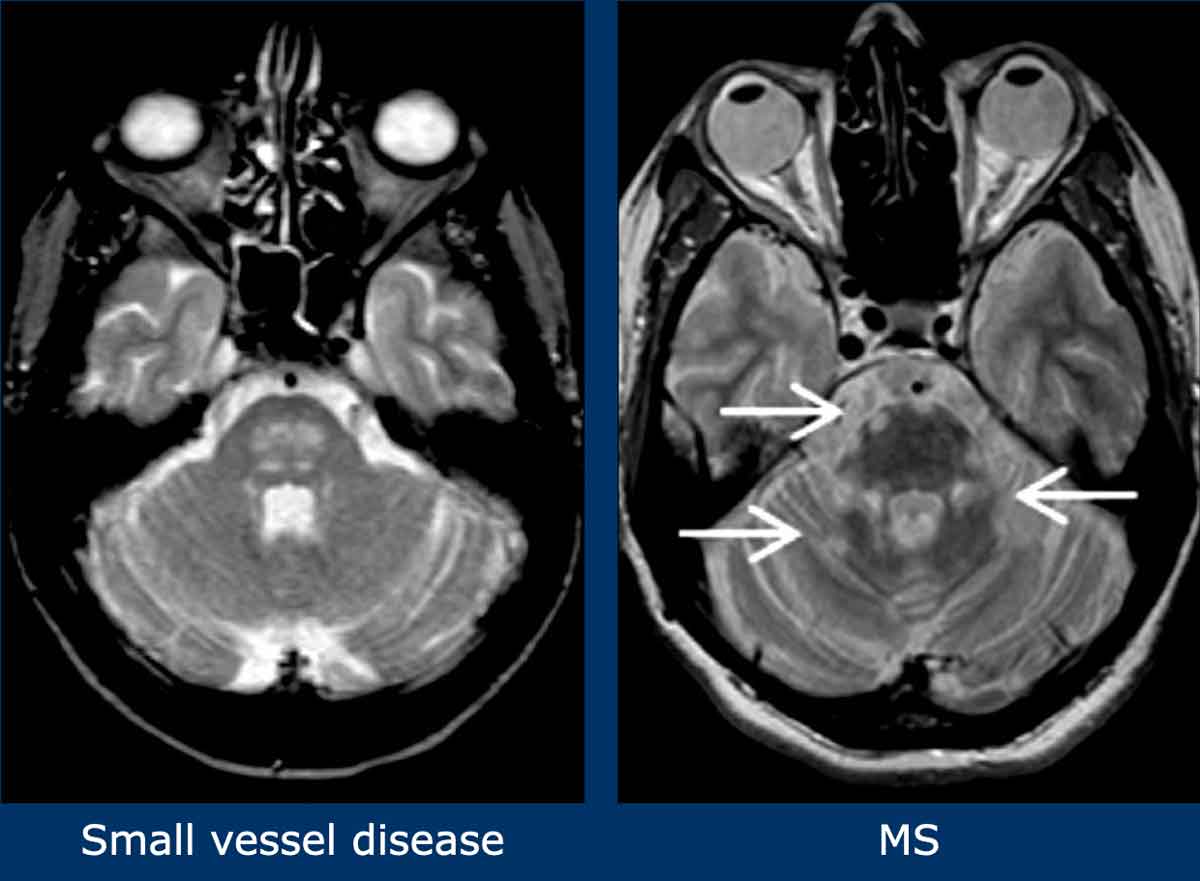
Brainstem lesions
In MS brainstem lesions are typically peripheral.
In small vessel disease the brainstem involvement is usually symmetrical and central.
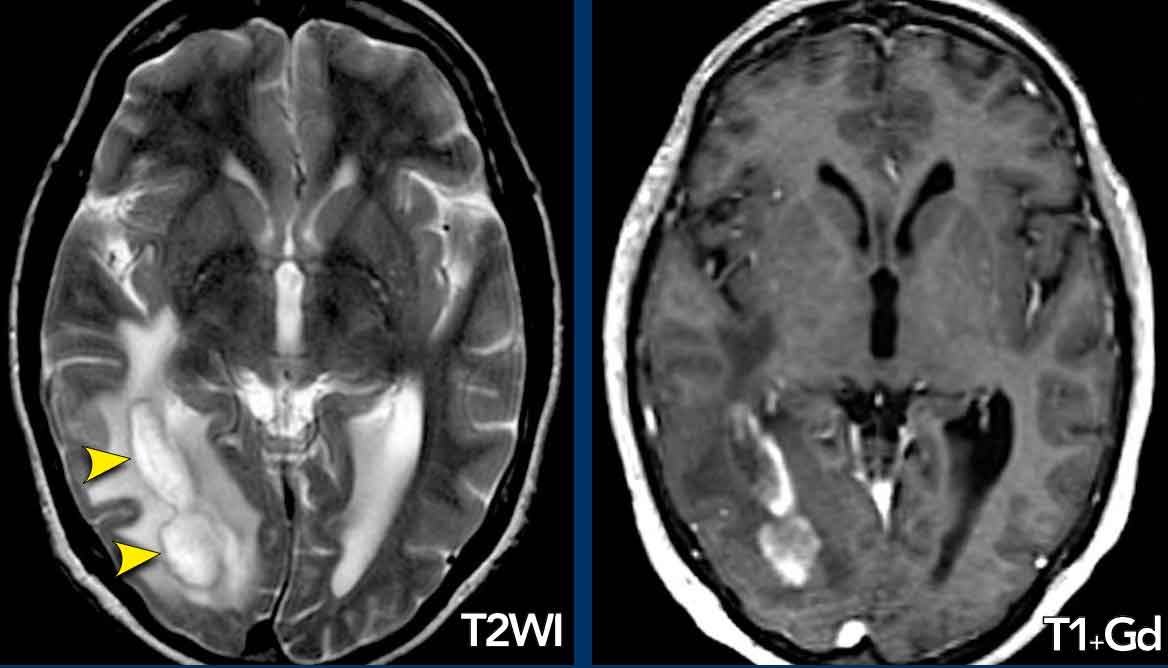
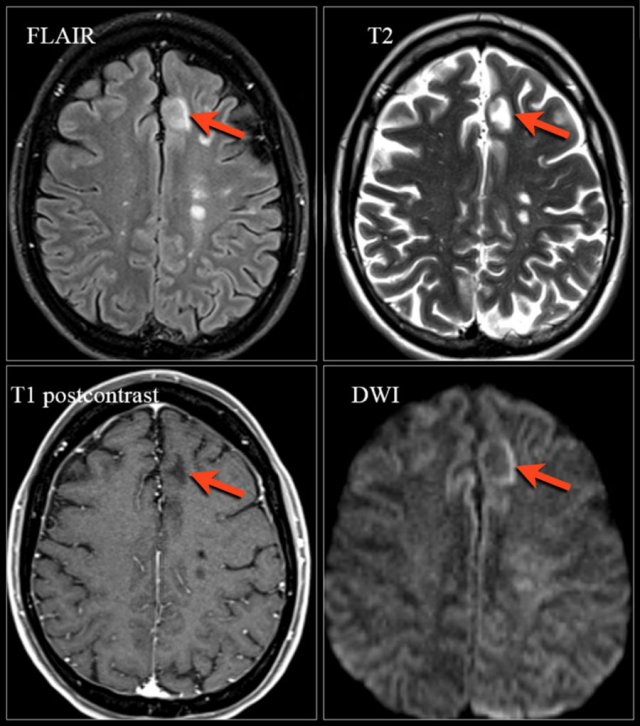
Enhancement
Enhancement is a characteristic feature of MS.
These images show a patient reassessed 3 months after the initial clinical attack.
Key MS findings in this case include:
- Multiple enhancing lesions
- Many lesions abutting the cortex, indicating U-fiber involvement
- All enhancing lesions are new, as gadolinium enhancement typically persists for only about 1 month, confirming dissemination in time
Perilesional edema resolves over time, leaving only the lesion core as a hyperintense area on T2-weighted imaging.
Tumefactive MS
Tumefactive multiple sclerosis is a rare MS variant characterized on MRI by large intraparenchymal lesions with disproportionately mild mass effect.
These lesions may exhibit peripheral enhancement, often with an incomplete ring—unlike the closed-ring enhancement typical of gliomas or abscesses.
Case Presentation
A 39-year-old male presented with subacute hemianopsia.
T2-weighted and post-gadolinium T1-weighted MRI revealed an intraparenchymal mass in the right temporal and occipital lobes, featuring a hypointense T2 rim and partial (open-ring) enhancement.
Surrounding edema was present, but mass effect was minimal.
Biopsy confirmed a demyelinating lesion.
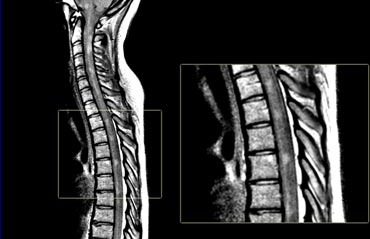
Spinal cord lesions
- Location/Size: Typically small, peripheral, and most common in the cervical cord; usually span fewer than 2 vertebral segments.
- Diagnostic Clue: Presence of a spinal cord lesion combined with a lesion in the cerebellum or brainstem strongly suggests MS.
- Differential Diagnosis: Spinal cord lesions are rare in most CNS diseases, except for ADEM, sarcoidosis, MOGAD and neuromyelitis optica spectrum disorder (NMOSD).
Spinal cord MRI is also valuable in adults older than 50 years and people with cerebrovascular pathology, as age-related or vascular white matter abnormalities that mimic MS are rarely associated with spinal cord lesions. - Prognosis: the presence of spinal cord lesions portends a poor prognosis
The images show multiple short-segment lesions in the spinal cord.
On PDW-images the spinal cord has a uniformly low signal intensity (like CSF), which gives the MS lesions a good contrast against the surrounding CSF and normal cord tissue.
Use a 512 matrix and cardiac gating for optimal results.
A good alternative for PDW is STIR or PSIR.
Make sure to have two different sagittal sequences and axial T2 with limited T2 weighting (avoid CISS and similar myelographic sequences).
MS Differential diagnosis
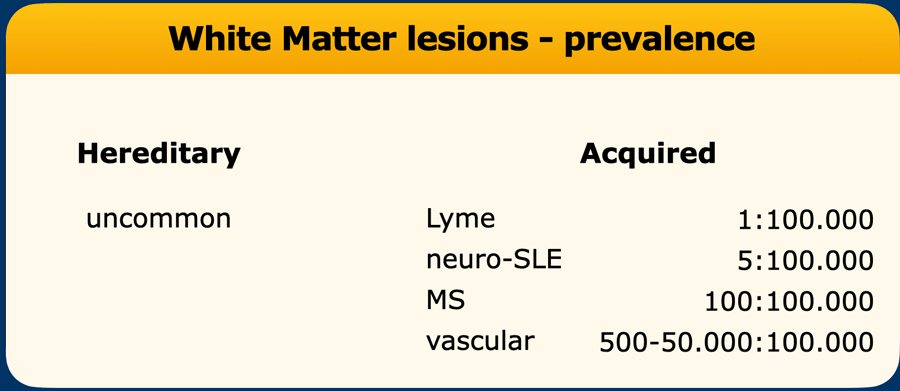
The differential diagnosis for multiple sclerosis (MS) is extensive, encompassing nearly all white matter diseases. While a comprehensive list exists, this discussion focuses on a select few, guided by practical rules:
- Clinical Suspicion + Multiple WMLs
If MS is clinically suspected and multiple WMLs are present, the primary differential is MS versus small vessel disease. - Clinical Suspicion + Supportive MRI-findings
If MS is clinically suspected and MRI findings support the diagnosis, uncommon alternatives need not be considered. - No Clinical Suspicion + Incidental WMLs
Do not include MS in the differential diagnosis if:- The clinician does not suspect MS.
- MRI shows deep WMLs only, without MS-characteristic features.
- Rationale: Vascular WMLs are 50–500 times more likely than MS plaques.
Small vessel disease
The differences between small vessel disease and MS are summarized in the table.
Typical for MS is involvement of corpus callosum, U-fibers, temporal lobes, brainstem, cerebellum and spinal cord.
This pattern of involvement is uncommon in other diseases.
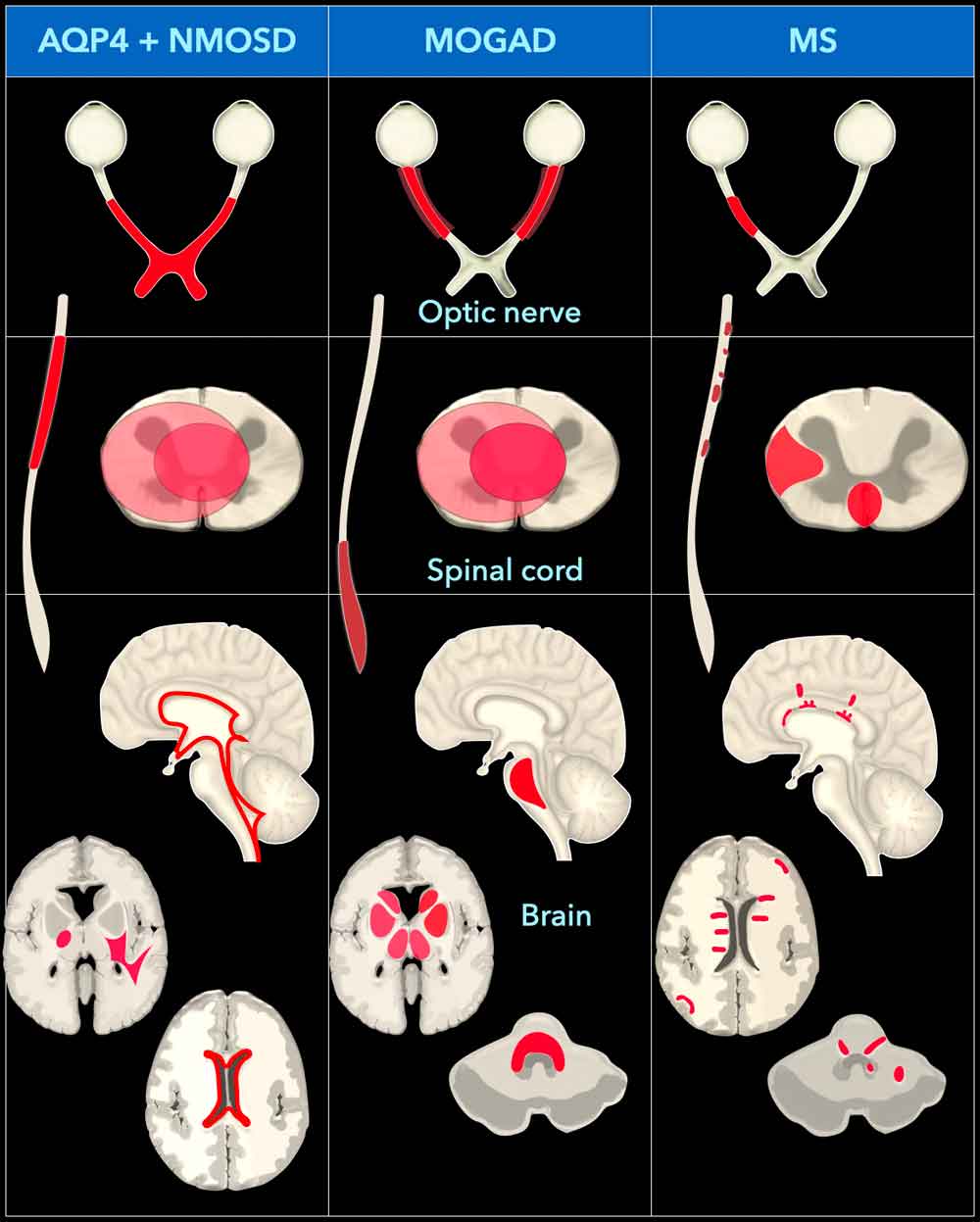
 Patient with NMOSD showing a longitudinally extensive cord lesion with marked swelling. Right-sided neuritis optica. The diagnosis was confirmed by an AQP4-AB titer of 1:1024.
Patient with NMOSD showing a longitudinally extensive cord lesion with marked swelling. Right-sided neuritis optica. The diagnosis was confirmed by an AQP4-AB titer of 1:1024.
AQP4 + NMOSD
Aquaporin-4 antibody-positive Neuromyelitis Optica Spectrum Disorder is a rare, autoimmune CNS disorder mediated by antibodies targeting the aquaporin-4 water channel on astrocytes.
Key Differential: Consider in patients presenting with severe or bilateral optic neuritis and extensive transverse myelitis.
Diagnosis: Confirmed by serum AQP4-IgG antibody detection.
Imaging Findings
- Spinal Cord: Longitudinally extensive transverse myelitis (≥3 vertebral segments), with T1-hypointensity and cord swelling. Lesions typically involve the central gray matter.
- Brain: Few T2-hyperintense lesions, but sometimes large hemispheric lesions. Involvement of medulla and area postrema (with hiccups)
- Distinction from MS: NMOSD lesions are centrally located, unlike the smaller, peripheral MS lesions.
MOGAD
Myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) is an autoimmune demyelinating disorder of the central nervous system, mediated by antibodies against myelin oligodendrocyte glycoprotein (MOG) and presents with:
- Optic neuritis: bilateral or long-segment involvement, perineural enhancement common.
- Transverse myelitis: extensive as in AQP4.
- Acute disseminated encephalomyelitis (ADEM)-like episodes.
- Brainstem/cerebral involvement; lesion often larger and more edematous than MS.
Diagnosis: confirmed by antibody testing (MOG-IgG) in serum or CSF.
Active spinal cord lesions in AQP4 + NMOSD and MOGAD tend to be longitudinally extensive (>3 vertebral segments) and are ill-defined with associated mass effect. Enhancement can be (open) ring or patchy
Involvement of the conus is suggestive of MOGAD.
On axial T2- and T2*-weighted images, MS spinal cord lesions are often round or wedge-shaped, involve the peripheral white matter, and might extend into the central grey matter.
By contrast, lesions in AQP4 + NMOSD and MOGAD are located centrally in the cord, spare the peripheral white matter, and might even be limited to the central grey matter— the so-called H-sign - seen especially in MOGAD in the acute phase.
In AQP4 + NMOSD, there might be cyst-like components, referred to as bright spotty lesions.
This diagram illustrates the distinctive imaging features of optic neuritis, myelitis, and brain lesions in AQP4-IgG–positive NMOSD patients versus MOG-IgG–positive NMOSD patients and multiple sclerosis patients.
It was adapted from the article:
Neuromyelitis Optica Spectrum Disorders: Spectrum of MR Imaging Findings and Their Differential Diagnosis by Bruna Garbugio Dutra [4].
ADEM
Acute Disseminated Encephalomyelitis (ADEM) is an immune-mediated demyelinating disorder, often post-infectious or post-vaccination, predominantly affecting children.
ADEM is a key differential for MS, distinguished by its monophasic course and characteristic imaging pattern.
Many cases are associated with MOG antibodies and now considered to be part of MOGAD.
MRI Findings
- Diffuse, relatively symmetrical supratentorial and infratentorial white matter lesions, frequently enhancing simultaneously.
- Preferential involvement of cortical gray matter and deep gray matter (basal ganglia, thalami).
Example
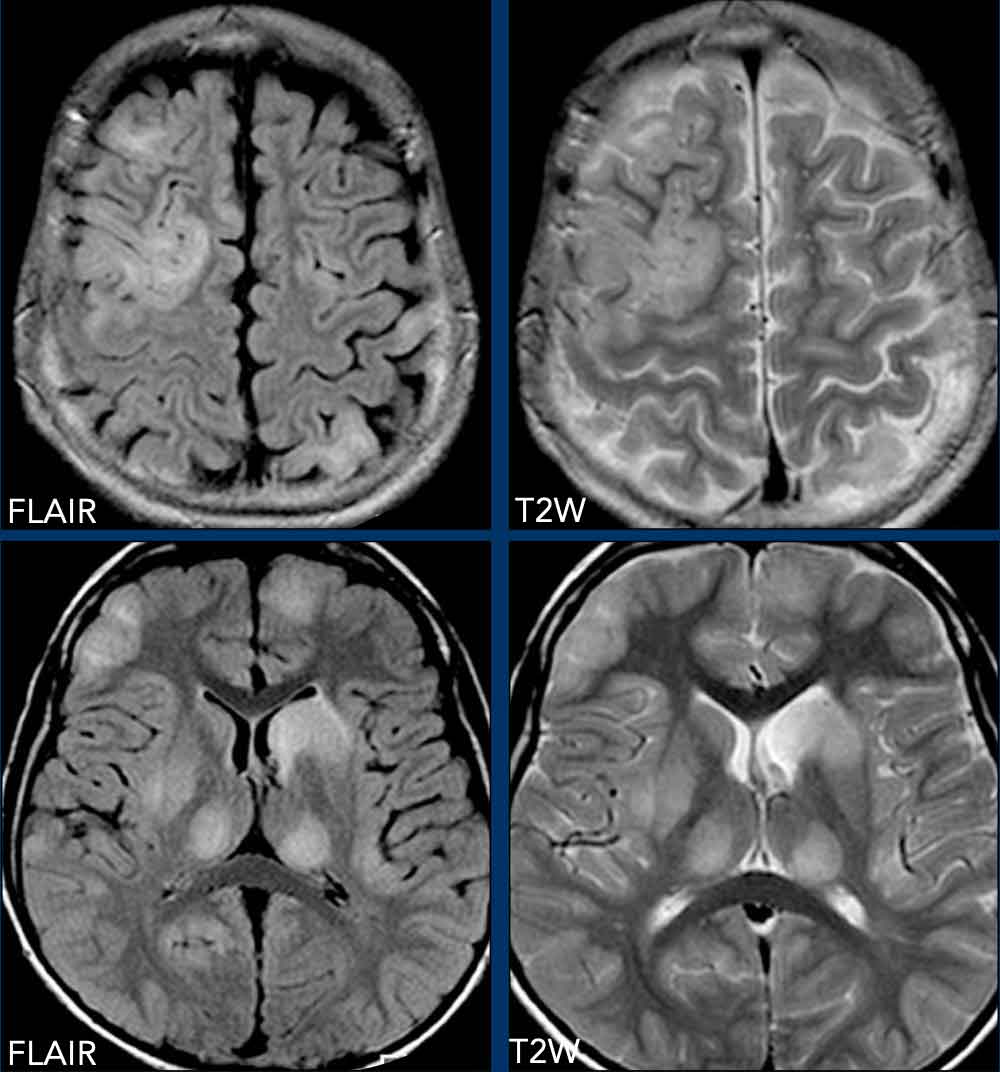
Axial FLAIR and T2W images demonstrate extensive cortical and deep gray matter involvement, including the thalamus.
The differences between ADEM and MS are summarized in the table.
 Natalizumab-associated PML. Images were kindly provided by Bénédicte Quivron CH Jolimont, La Louvière, Belgium
Natalizumab-associated PML. Images were kindly provided by Bénédicte Quivron CH Jolimont, La Louvière, Belgium
Natalizumab-associated PML
Natalizumab, a monoclonal antibody targeting α4-integrin, is approved for relapsing-remitting MS due to its efficacy in reducing clinical relapses and MRI activity.
A rare but severe adverse effect is an increased risk of PML.
Progressive Multifocal Leukoencephalopathy (PML) is a demyelinating disease caused by JC virus reactivation in immunosuppressed patients.
Diagnosis of PML is based on:
- Clinical presentation (subacute neurological deficits)
- Detection of JC virus DNA in CSF
- Characteristic MRI findings
MRI Features of Natalizumab-Associated PML
- Location:
- Supratentorial subcortical white matter (especially U-fibers and cortical gray matter)
- Less commonly: posterior fossa, deep gray matter
- Signal Characteristics:
- T2-hyperintense (occasionally with satellite lesions)
- T1-isointense or hypointense (depending on demyelination severity)
- Contrast enhancement (~30% of cases)
- DWI hyperintensity (particularly at lesion borders, indicating active infection and white matter swelling)
Note: Imaging findings in natalizumab-associated PML are more heterogeneous and fluctuating compared to classic HIV-associated PML. Early recognition is critical for management.
Distinguishing progressive MS from natalizumab-associated PML can be challenging.
Refer to the table for key imaging differences.
Optimal MRI Protocol for Natalizumab-Associated PML:
- FLAIR: highest sensitivity for detecting of PML lesions
- T2-weighted sequences: reveals lesion details (e.g., microcysts).
- T1 with and without contrast: Assesses demyelination severity and inflammatory activity.
- DWI is useful for the detection of active infection.
Further information: www.MS-PML.org
MRI protocol
Brain Imaging
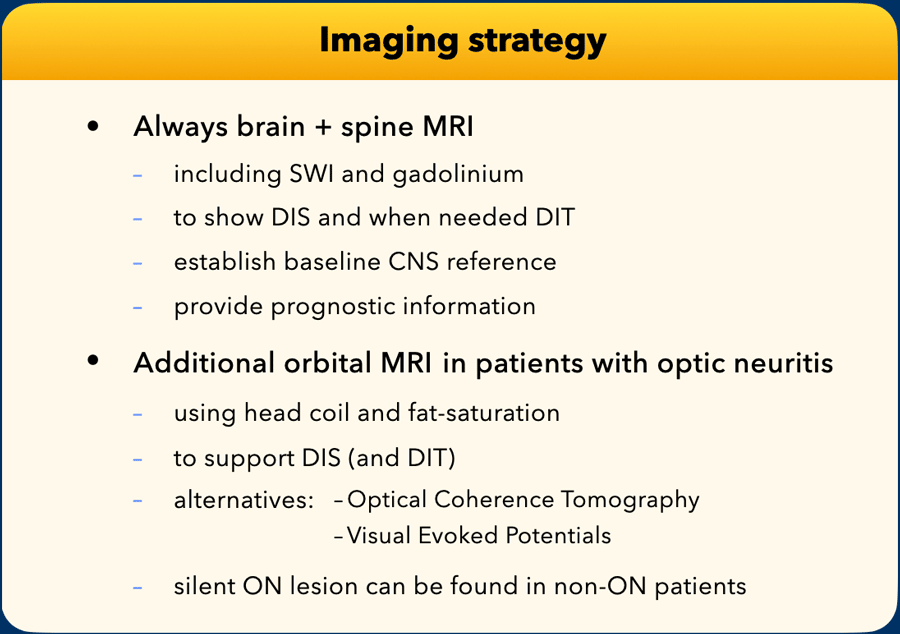
In patients suspected of MS, we recommend brain and whole spinal cord MRI to confirm diagnosis, exclude differentials, inform prognosis, and provide a baseline for follow-up comparisons.
Intravenous gadolinium should be administered to detect enhancing lesions and aid differential diagnosis.
The presence of both contrast-enhancing and non-enhancing lesions may fulfill criteria for dissemination in time.
Spinal Cord Imaging
Spinal cord MRI is routinely indicated at diagnosis, as spinal cord lesions contribute to DIS and indicate a poorer prognosis. It is especially valuable in patients with spinal cord symptoms to confirm demyelination and exclude alternative causes.
In adults over 50 or those with cerebrovascular disease, spinal cord MRI helps distinguish MS from age-related or vascular white matter changes, which rarely involve the spinal cord.
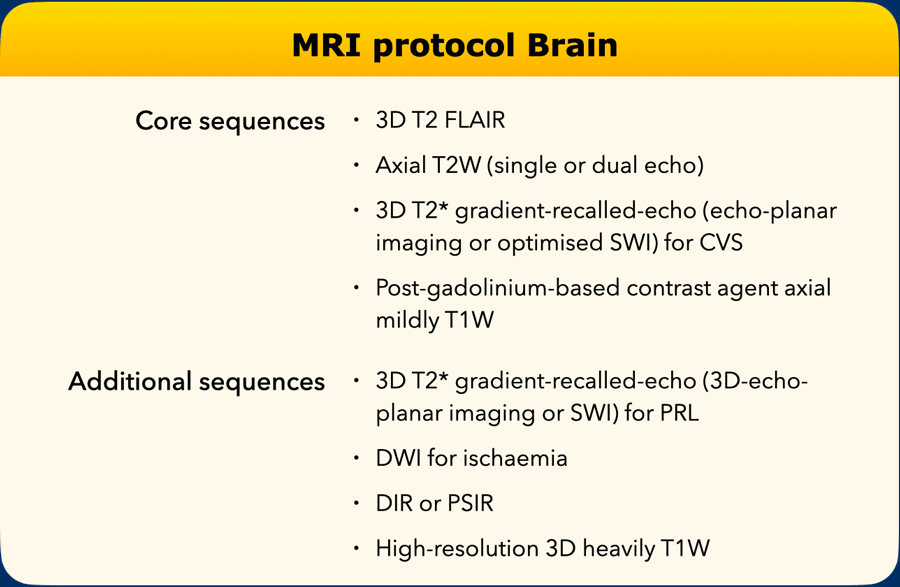
Brain protocol
Preferred settings:
- Field strength ≥ 1·5 T
- Acquisition: 3D preferred
- Slice thickness: 3D: 1 mm isotropic. 2D: ≤ 3 mm, no gap
- In-plane resolution: ≤ 1 mm × 1 mm
- Coverage: Whole brain
- Axial scan orientation: Subcallosal or AP commissure plane
Gadolinium is administered at the start of the examination because the longer you wait the more enhancement you will see on the T1W images, which are made about 15 minutes later.
FLAIR is ideal for detection of lesions in the corpus callosum and allows for better detection of smaller and juxtacortical lesions.
The T2W scan can be turbo/fast spin-echo and help confirm thalamic and infratentorial lesions.
Spinal cord Protocol
Note
Do not use FLAIR or myelographic sequences (very heavily T2-weighted, eg, CISS) as they will only demonstrate 10% of the lesions.
Optic nerve protocol
Optic nerve MRI sequences, required if there is an initial presentation of optic neuritis, include at minimum coronal fat-suppressed T2-weighted or short tau inversion recovery sequences.
When gadolinium-based contrast agents are given in cases of acute optic neuritis, post-gadolinium-based contrast agents coronal fat-suppressed T1-weighted sequences are recommended.
Coverage should include the optic nerve and the optic chiasm, with coronal planes oriented perpendicular to the optic nerve. When axial images are added, they should align with the optic nerve and chiasm.
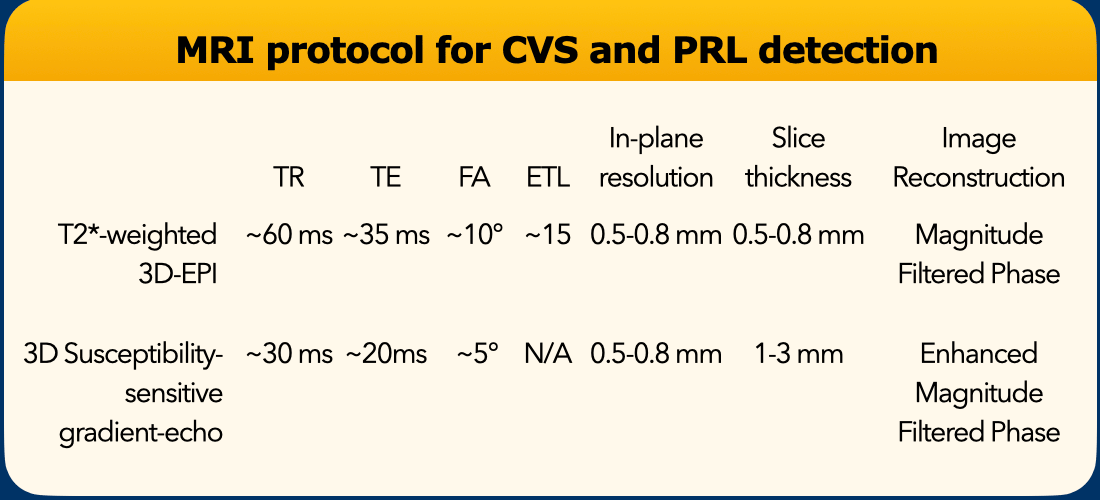
CVS and PRL protocol
Central vein sign (CVS) and paramagnetic rim lesions (PRL) can be detected on various gradient-echo MRI sequences, like SWI, SWIp, SWAN, SWAN-venule or similar sequences, providing that there is sufficient susceptibility (or T2*) weighting and maximum 1·0 mm isotropic spatial resolution. Make sure to save phase maps that are better to detect PRL.
One highly efficient approach uses 3D segmented echo-planar imaging.
Administration of gadolinium-based contrast agents immediately before or during acquisition of susceptibility-sensitive sequences improves detection of the CVS.